For each of the following compounds, determine whether the ring is activated or deactivated, then determine the
Question:
For each of the following compounds, determine whether the ring is activated or deactivated, then determine the strength of activation/deactivation, and finally, determine the expected directing effects.
(a)
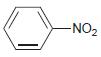
(b)
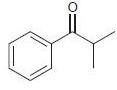
(c)
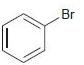
(d)
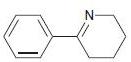
(e)
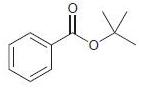
(f)
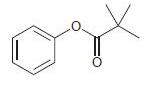
Transcribed Image Text:
-NO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (17 reviews)
a The nitro is strongly deactivating and metadirecting b An acyl group ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds determine whether it exhibits a cis configuration or a trans configuration or whether it is simply not stereo-isomeric. a. b. c. d. e. f. g. F
-
For each of the following compounds, draw the important resonance forms. Indicate which structures are major and minor contributors or whether they have the same energy. (a) H2CNN (b) H2C=CH-NO2 (c)...
-
For each of the following compounds, give the systematic name and the common name (for those that have common names), and indicate whether the amines are primary, secondary, or tertiary: a. b. c....
-
For rigid-body motion, the strains will vanish. Under these conditions, integrate the straindisplacement relations (7.6.1) to show that the most general form of a rigid-body motion displacement field...
-
What is a unique selling proposition? What role should it play in a company's advertising strategy?
-
Larry Kraft plans to open six more restaurants during the next five years. He believes that he will then have 125 employees. a. What recommendations would you make for Larry at this time? b. Could...
-
In Example 2, the adult weighing 285 pounds decides to not participate in the study. What is the median weight of the remaining adults? Data from Example 2 Find the median of the weights listed in...
-
Grant Company purchased the net assets of Harding Company on January 1, 2015, and made the following entry to record the purchase: Provide the correct title for the $40,000 credit account above and...
-
MCO Leather manufactures leather purses. Each purse requires 2 pounds of direct materials at a cost of $3 per pound and 0.7 direct labor hour at a rate of $14 per hour. Variable overhead is budgeted...
-
Empirical research has found that there have been significant increases in stock price on the day an initial dividend (i.e., the first time a firm pays a cash dividend) is announced. What does this...
-
Predict and explain the regiochemical outcome for chlorination of bromobenzene.
-
The following compound has two aromatic rings. Identify which ring is expected to be more reactive toward an electrophilic aromatic substitution reaction.
-
Wilton, AG had net sales in 2019 of 1,400,000. At December 31, 2019, before adjusting entries, the balances in selected accounts were Accounts Receivable 250,000 debit, and Allowance for Doubtful...
-
A relatively costly form of life insurance protection that includes a savings feature. a. malpractice b. professional c. term ins urance d. title insurance e. whole life insurance
-
Suppose a companys return on invested capital is less than its WACC. What happens to the value of operations if the sales growth rate increases? Explain your answer.
-
A form of insurance that assures the buyer of real property that there are no other claims to the ownership. a. malpractice b. professional c. term ins urance d. title insurance e. whole life...
-
At year-end 2018, Wallace Landscapings total assets were $2.17 million, and its accounts payable were $560,000. Sales, which in 2018 were $3.5 million, are expected to increase by 35% in 2019. Total...
-
Start with the partial model in the file Ch09 P10 Build a Model.xlsx on the textbooks Web site, which contains the 2018 financial statements of Zieber Corporation. Forecast Ziebers 2019 income...
-
Sketch the graph of an appropriate function and then use the graph to solve each equation or inequality. First determine whether the expression is an equation or an inequality. When appropriate, use...
-
Explain why each of the following is either a private good or a public good: traffic lights, in line skates, a city park, a chicken salad sandwich, a tennis racket, national defense, a coastal...
-
Two of the compounds given in Fig. Pl 1.78 form epoxides readily when treated with -OH, one forms an epoxid slowly, and one does not form an epoxide at all. Identify the compound(s) in each category...
-
Two of the compounds given in Fig. Pl 1.78 form epoxides readily when treated with -OH, one forms an epoxid slowly, and one does not form an epoxide at all. Identify the compound(s) in each category...
-
Two of the compounds given in Fig. Pl 1.78 form epoxides readily when treated with -OH, one forms an epoxid slowly, and one does not form an epoxide at all. Identify the compound(s) in each category...
-
An all-equity firm is considering the purchase of a depreciable asset that costs $680,000 and would be fully depreciated over six years using the straight-line method. The asset is expected to...
-
A firm owns three department stores. Each store has a debt-equity ratio of 16 percent and makes interest payments of $44,000 at the end of each year. The cost of the firm's levered equity is 18...
-
Belgian Bite Corp. is considering a project that has the following cash flow data. What is the project's IRR? Year 0 1 2 3 Cash flows $1,100 $450 $470 $490

Study smarter with the SolutionInn App


