For each of the following pairs of compounds, determine the relationship between the two compounds: a. b.
Question:
a.
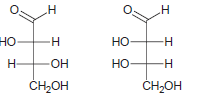
b.
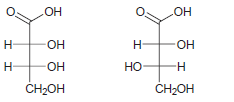
c.
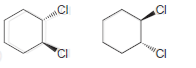
d.
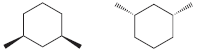
e.
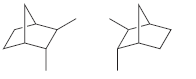
f.
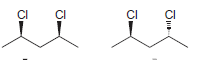
g.
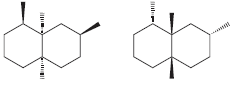
h.
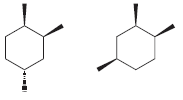
i.
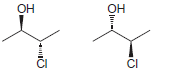
j.
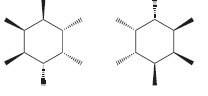
k.
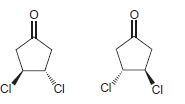
l.
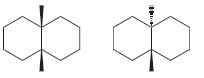
Transcribed Image Text:
н н но- —н Но— —н но- Н -ОН -н CH-он ČH2OH он но" н- -ОН н- он Н ОН Но- н CH-ОН ČH2OH エ エ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (12 reviews)
a Diastereomers b Diastereomers c En...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following pairs of compounds undergoes a Bronsted acid-base reaction for which the equilibrium lies to the right. Give the products of each reaction, and identify the acid, the base, the...
-
In each of the following pairs of compounds one is chiral and the other is achiral. Identify each compound as chiral or achiral, as appropriate. a. b. c. d. CICH CHCH OH and HOCH CHCH,OH CI OH CH,CH...
-
In each of the following pairs of compounds choose which one will react faster with the indicated reagent, and write a chemical equation for the faster reaction: (a) Toluene or chlorobenzene with a...
-
Based on Exhibit 1, the expected future value of Bond I at maturity is closest to: A. 98.80. B. 103.74. C. 105.00. Lena Liecken is a senior bond analyst at Taurus Investment Management. Kristel...
-
Why are pro forma financial statements important to the financial planning process?
-
Asaya Clothing Inc. is a manufacturer of winter clothes. It has a Knitting department and a Finishing department. This exercise focuses on the Finishing department. Direct materials are added at the...
-
True or false: RMS stands for relative mean squared.
-
On July 15, Mann Company sold $600,000 in accounts receivable for cash of $500,000. The factor withheld 10% of the cash proceeds to allow for possible customer returns or account adjustments. An...
-
The number of active Facebook users hit 2.414 billion at the end of June 2019 and 2.498 billion at the end of December 2019. With in months since the start of 2019, let f(r) be the number of active...
-
A 30-m steel tape measured 29.991 m when standardized fully supported under a 5.500-kg pull at a temperature of 20C. The tape weighed 1.22 kg and had a cross-sectional area of 0.016 cm2. What is the...
-
Why is the EOQ model described as robust?
-
Identify the stronger nucleophile: (a) NaSH vs. H 2 S (b) Sodium hydroxide vs. water (c) Methoxide dissolved in methanol vs. methoxide dissolved in DMSO
-
Assume the photodiode of this problem as an ideal structure with \(100 \%\) quantum efficiency and area \(1 \mathrm{~cm}^{2}(=1 \mathrm{~cm} \times 1 \mathrm{~cm})\). In addition, assume it is...
-
Explain how the law of diminishing returns provides a natural tendency toward competitive balance.
-
Indicate whether each of the following statements is true or false by writing T or F in t he a nswer c olumn. A straight bill of lading is an instrument used to collect money owed.
-
While football and baseball teams have gone from multipurpose to football- and baseball-only facilities, basketball and hockey teams continue to share arenas. Why?
-
If a majority of people do not want a stadium built in their town, how can building it lead to an improvement in social well-being?
-
Who will benefit most from the new stadium in Minneapolis, the Vikings, their fans, or the city as a whole?
-
Solve each problem The force of the wind blowing on a vertical surface varies jointly as the area of the surface and the square of the velocity. If a wind of 40 mph exerts a force of 50 lb on a...
-
With your classmates, form small teams of skunkworks. Your task is to identify an innovation that you think would benefit your school, college, or university, and to outline an action plan for...
-
Ethyl triflate is much more reactive than ethyl mesylate toward nucleophiles in SN2 reactions. (a) Give the structures of all of the products formed when each compound reacts with potassium iodide in...
-
How many grams of CrO3, are required to oxidize 10 g of 2-heptanol to the colresponding ketone?
-
How many grams of CrO3, are required to oxidize 10 g of 2-heptanol to the colresponding ketone?
-
Discuss the challenges and opportunities of scaling up process intensification concepts from laboratory-scale experiments to industrial-scale applications, addressing issues related to...
-
Suppose the pressure in the esophagus is - 1.95 mm Hg while that in the stomach is + 18.5 mm Hg. To what height, in centimeters, could stomach fluid rise in the esophagus, assuming it has a density...
-
When light of wavelength190nm falls on acesiumsurface, electrons having a maximum kinetic energy of 4.39eV are emitted. Find values for the following. (a) the work function of cesium eV? (b) the...

Study smarter with the SolutionInn App


