Identify the major and minor products for the E2 reaction that occurs when each of the following
Question:
Identify the major and minor products for the E2 reaction that occurs when each of the following substrates is treated with a strong base:
a.
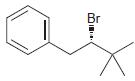
b.
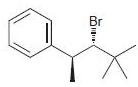
c.
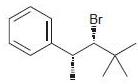
d.
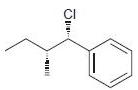
e.
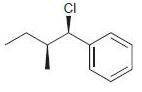
f.
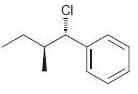
g.
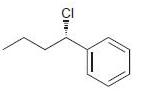
h.
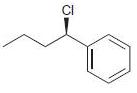
Transcribed Image Text:
Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
a b c...View the full answer

Answered By

Ishrat Khan
Previously, I have worked as an accounting scholar at acemyhomework, and have been tutoring busines students in various subjects, mostly accounting. More specifically I'm very knowledgeable in accounting subjects for college and university level. I have done master in commerce specialising in accounting and finance as well as other business subjects.
5.00+
134+ Reviews
427+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When menthyl chloride is treated with a strong base, only one elimination product is observed. Yet, when neomenthyl chloride is treated with a strong base, two elimination products are observed. Draw...
-
When (1R, 2R)-2-bromocyclohexanol is treated with a strong base, an epoxide (cyclic ether) is formed. Suggest a mechanism for formation of the epoxide: Strong base Br An epoxide
-
Identify the major and minor products for each of the following E2 reactions: a. b. c. d. e. f. CI NaOEt CI t-BUOK
-
Discuss the role of intrinsic & extrinsic cues in the perceived quality of you product/service(Unilever company; Lipton tea as an example). Include it in the about section of assignment
-
Draw a phase diagram and begin to fill in the blanks: A soil specimen has total volume of 80,000 mm3 and weighs 145 g. The dry weight of the specimen is 128 g, and the density of the soil solids is...
-
Repeat Experiment 6.2 using \(F_{\mathrm{s}}=10 \mathrm{~Hz}\). Compare the magnitude response of the resulting discrete-time transfer function with the one obtained in the experiment. Experiment 6.2...
-
The dataset ToyotaCorolla.jmp contains data on used cars on sale during the late summer of 2004 in the Netherlands. It has 1436 records containing details on 38 attributes, including Price, Age,...
-
Suppose you know that a companys stock currently sells for $75 per share and the required return on the stock is 11 percent. You also know that the total return on the stock is evenly dividends...
-
A bond with a $1,000 face value and 13 years to maturity is currently selling for $920. If the yield to maturity is 5.6%, what is the coupon rate of the bond?
-
Ten sources, six with a bit rate of 200 kbps and four with a bit rate of 400 kbps, are to be combined using multilevel TDM with no synchronizing bits. Answer the following questions about the final...
-
Identify the major and minor products for the E2 reaction that occurs when each of the following substrates is treated with a strong base: a. b. c. d. e. f. g. h. Br
-
Identify an alkyl halide that could be used to make the following alkene:
-
Financial statements all have a goal. The cash flow statement does as well. Describe how the statement of cash flows helps investors and creditors perform each of the following functions: a. Predict...
-
Write named CHECK constraints for the customer balance is greater than or equal to zero and customer state is one of CO or CA in the CREATE table statement to add the named CHECK constraints ?
-
A copy machine cost $39,000 when new and has accumulated depreciation of $25,000. Suppose Print Center sold the machine for $14,000. What is the result of this disposal transaction?
-
In a city water-filtration plant, it was desired to filter 5000 gal of water per hour through a sand filter bed to remove all the suspended matter and solids from the water. A vertical...
-
For the reaction 2HI(g) H(g) +12(g), it was found that the rate of HI to be R = 5.0 10 mmolHI -1 HI *I**s* what is (a) the average rate of formation of H in the same reaction and (b) the unique...
-
On June 2 0 , 2 0 1 1 , the Company . . . finalized the Company s acquisition from Unilever of the Sanex personal care business . . . for an aggregate purchase price of $ 9 6 6 . . . . This...
-
How soon will nanotechnology give rise to commercial products?
-
A red card is illuminated by red light. What color will the card appear? What if its illuminated by blue light?
-
Name the following alkynes, and predict the products of their reaction with (i) H2 in the presence of a Lindlar catalyst and (ii) H3O+ in the presence ofHgSO4: (b) (a)
-
From what alkyne might each of the following substances have been made? (Yellow-green =Cl) (b) (a)
-
How would you prepare the following substances, starting from any corn- pounds having four carbons orfewer? (a) (b)
-
Calendar Plc have a year end of 30/09/X2. They have prepared the following information about their non-current assets: Land was revalued to 4.2 million on 28/09/X2 All buildings are depreciated using...
-
Antique Accents reported the following on its 2020 income statement. Using the income statement and the balance sheet given below, prepare the statement of cash flows using the indirect method. For...
-
Jamal, Maggie, and Kendra are dividing an estate consisting of a house, a vacation home, and a small business. Their valuations are shown below. Determine the payment that Kendra receives from the...

Study smarter with the SolutionInn App


