Identify the reactants that you would use to make each of the following compounds: (a) (b) NH2
Question:
(a)
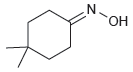
(b)
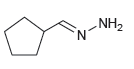
Transcribed Image Text:
он NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a b ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the reagents that you would use to convert β-d-glucopyranose into each of the following compounds: (a) (b) (c) (d) CH,OH - - - H H
-
Use Table 3.2 to make each of the following conversions. a. Convert US$350 to Canadian dollars. b. Convert C$200 to euros. c. Convert US$175 to Swiss francs. d. Convert 250 United Kingdom pounds to...
-
Identify whether each of the following compounds can be made using a direct Friedel-Crafts alkylation or whether it is necessary to perform an acylation followed by a Clemmensen reduction to avoid...
-
Suggest a good synthetic method for preparing each of the following haloalkanes from the corresponding alcohols. CH3 H3C CI I () CH-CH-CH,CI (b) CH;CH,CHCH,Br () (d) CH;CHCH(CH3)2
-
Prepare another infusion Center Capacity Level Forecast as follows: Assume the same three infusion chairs, but add another nurse for either four or six hours per day. How would this change the daily...
-
Why is it difficult to identify and describe an organizations culture?
-
A certain underlying state graph is a tree where each node has three successor nodes, indexed \(a,b, c\). There are two assets defined on this tree which pay no dividends except at the terminal time...
-
In the following figure, a consumer is initially in equilibrium at point C. The consumers income is $ 400, and the budget line through point C is given by $ 400 $ 100X + $ 200Y. When the consumer is...
-
How do matrix organizational structures balance the dual imperatives of functional specialization and project-based collaboration, and what challenges do they pose in terms of role clarity and...
-
Starr Scopes, Inc., produces telescopes for use by high school students. All direct materials used in the production of telescopes are added at the beginning of the manufacturing process. Labor and...
-
A sample of students from our statistics classes reported their GPAs, indicated their genders, and stated whether they were in the universitys Greek system (i.e., in a fraternity or sorority)....
-
Draw a plausible mechanism for each of the following reactions: (a) (b) [H2SO4] Et,NH 'N' -H20 [H2SO4] Me-NH -H20
-
A Ltd. is committed to supply 24,000 bearings per annum to B Ltd. on a steady basis. It is estimated that it costs 10 paise as inventory holding cost per bearing per month and that the set-up cost...
-
After failing to complete a merger in the three prior attempts noted in the case, why should the proposed transaction be successful this time? What is different? Is this a good or a bad thing?
-
Discuss five intermediation services performed by financial intermediaries when direct financial claims are being transformed into indirect financial claims.
-
In the early 2000's, St. Paul - Travelers decided to sell one of its subsidiary groups, Geovera Holdings, to a private equity firm.After the sale, A.M. Best downgraded the financial strength ratings...
-
What condition would cause capital structure management to be meaningless activity? Why should the financial manager be familiar with the business cycle?
-
Each week you will compose and post an entry in what you should view as a continuing Learning Journal for the course. In your weekly entry, focus on those assigned works that were not the focus of...
-
A bean bag is randomly thrown onto the square table top shown below and does not touch a line. Determine the probability that the bean bag lands on A red or yellow area.
-
31. What is the income that can be received over 15 years from $500,000 earning 6% annually? 32. What is the semiannual payment required to retire $50,000 in debt over 5 years at 8% compounded...
-
In infrared spectra, the carbonyl group is usually indicated by a single strong and sharp absorption. However, in the case of carboxylic acid anhydrides, two peaks are observed even though the two...
-
Sulfur dioxide (SO2) has a dipole moment ( = 1.63 D); on the other hand, carbon dioxide has no dipole moment ( = 0 D). What do these facts indicate about the geometry of sulfur dioxide?
-
Write structural formulas for four compounds with the formula C3H6O and classify each according to its functional group. Predict IR absorption frequencies for the functional groups you have drawn.
-
Complete the Price, Advertising, and Profitability chart. a . ?Notice that the series for $ 2 5 , 0 0 0 , 0 0 0 ?is already on the chart. b . ?Add the series for advertising budgets $ 5 0 , 0 0 0 , 0...
-
Solve for the missing information designated by "?" in the following table. (Use 365 days in a year. Round the inventory turnover ratio to one decimal place before computing days to sell. Round days...
-
Grove Media plans to acquire production equipment for $800,000 that will be depreciated for tax purposes as follows: year 1, $320,000; year 2, $180,000; and in each of years 3 through 5, $100,000 per...

Study smarter with the SolutionInn App


