Identify which of the following monomers would be most reactive toward cationic polymerization. OAc CN CI
Question:
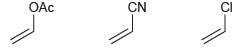
Transcribed Image Text:
OAc CN CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
least r...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify which of the following monomers would be most reactive toward anionic polymerization: CH3 OAc CN
-
Which of the following monomers might you expect would lead to a conducting polymer? a. b. H2C==CHCH3 --,
-
Which of the following monomers might you expect to lead to a conducting polymer? a. CH3CH==CHCH3 b. CH CC CH
-
Bruceton Farms Equipment Company had goodwill valued at $80 million on its balance sheet at year-end. A review of the goodwill by the company's CFO indicated that the goodwill was impaired and was...
-
Use the following information for The Windshield Helper, a retail merchandiser of auto windshields, to compute the cost of goods sold: Web Site Maintenance...
-
How much would be appropriate for wealthy countries to contribute to global health? Why should we do more than we do now? What's in it for us?
-
The following data come from a study in which random samples of the employees of three government agencies were asked questions about their pension plan: Use the 0.01 level of significance to test...
-
The following relate to auditors' independence: Required a. Why is independence so essential for auditors? b. Compare the importance of independence of CPAs with that of other professionals, such as...
-
Demanding Nursing profession. It has become an especially challenging hiring and ongoing management category in recent years. Covid, overall workload demands, advanced education requirements, and...
-
Valu-Com Electronics manufactures five different models of telecommunications interface cards for personal and laptop computers. As summarized in the following table, each of these devices requires...
-
Nitroethylene undergoes anionic polymerization so rapidly that it is difficult to isolate nitroethylene without it polymerizing. Explain.
-
Draw a region of an alternating copolymer constructed from vinyl chloride and ethylene.
-
In Exercises 5 through 12, determine where the graph of the given function is concave upward and concave downward. Find the coordinates of all inflection points. f(x) = x 3 + 3x 2 + x + 1
-
Make a recommendation which two clinics should be closed, with a brief explanation why they should close and the others should remain open. Then, identify and describe in detail the consequences you...
-
Write reflection should tie all the material together based on a theme you identify instead of just focusing on one topic. In the reflection, you will also: Summarize key ideas from the readings,...
-
It is common in interviews to be asked what your short-term and long-term career goals are. At which point, it might be informative if it is shared that you would like to have an international...
-
How do Promotions and Compensation go hand and hand? Do people want opportunities to grow in their careers, if they don't find it in their organization will they find it elsewhere?
-
Describe 3 different scenarios where social and/or human capital have played a role in conflict or negotiation outcomes. In your most recent job, describe the degree of social and human capital you...
-
Cerberus Security Company produces a cash fl ow of $200 per year and is expected to continue doing so in the infinite future. The cost of equity capital for Cerberus is 20 percent, and the firm is...
-
Consider the circuit of Fig. 7.97. Find v0 (t) if i(0) = 2 A and v(t) = 0. 1 3 ett)
-
Give correct names for the following amines: (a) (b) (c) (d) (e) (f) NH, CH3 CHCHCH3 NHCH3 NH2 ik CH3 NH NH2 NH CHO
-
Show how you would accomplish the following synthetic conversions. (a) Benzyl bromide benzylamine (b) 1-bromo-2-phenylethane 3-phenylpropan-1-amine (c) Pentanoic acid pentan-1-amine (d) Pentanoic...
-
Show how you would prepare the following aromatic amines by aromatic nitration, followed by reduction. You may use benzene and toluene as your aromatic starting materials. (a) Aniline (b)...
-
Reflecting on the book Who Gets Promoted, Who Doesn't, and Why (Asher, 2008), answer the following questions in depth. Explain the statement, "all business is sales." What six things matter? If...
-
Reflecting on the book Who Gets Promoted, Who Doesn't, and Why (Asher, 2008), answer the following questions in depth. No matter what you have done in the past, the boss really doesn't care. Why?...
-
Do you think Richard Scrushy should have been found guilty or acquitted for his role in the case against HealthSouth? https://www.wsj.com/articles/SB111702610398942860

Study smarter with the SolutionInn App


