In each of the following cases draw the structure of an alkyl halide that will undergo an
Question:
a.
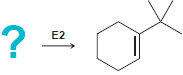
b.
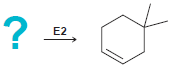
c.
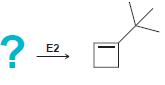
d.
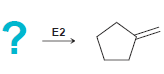
Transcribed Image Text:
? = E2 E2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each of the following cases, imagine that the two reactants shown are allowed to reirct,in the presence of alcohol dehydrogenase. Tell whether the ethanol formed is chiral. If the ethanol is...
-
Draw the alkoxide formed in each of the following cases: a. b. c. d. HO ? Na NaH
-
Draw the structure of an alkyl halide or carboxylic acid that might serve as a precursor in the preparation of each of the following amines: (a) (b) (c) `NH2 NH2
-
A magnet of mass 5 . 0 1 kg is suspended from the ceiling by a cord as shown. A large magnet is somewhere off to the right, pulling on the small hanging magnet with a constant force of F = 8 0 . 4 N...
-
Before approaching this assignment, be sure that you have watched the following video. Budgetary planning Featuring Babycakes *FULL VIDEO* (2016) YouTube Develop a paper in which you address the...
-
Repeat Exercise 9.9 for the lowpass analysis filter equal to \[H_{0}(z)=\frac{1}{4} z^{-5}+z^{-3}+z^{-2}+z^{-1}+2\] Exercise 9.9 Consider a perfect reconstruction filter bank which satisfies the QMF...
-
Provide an example for use case generalization. What is its significance?
-
Mike Lyons incorporated Lyons Concrete, Inc., in Montana, but did not file its first annual report, so the state involuntarily dissolved the firm in 1996. Unaware of the dissolution, Lyons continued...
-
The density of a fluid changes with depth from the surface h according to the following formula: (h) = ch 2 , where c is a constant. A. What an expression for the gauge pressure p as a function of...
-
Assume that we make an enhancement to a computer that improves some mode of execution by a factor of 10. Enhanced mode is used 50% of the time, measured as a percentage of the execution time when the...
-
Suppose that the paper clip industry is perfectly competitive. Also assume that the market price for paper clips is 2 cents per paper clip. The demand curve faced by each firm in the industry is: a....
-
A purely competitive firm whose goal is to maximize profit will choose to produce the amount of output at which: a. TR and TC are equal. b. TR exceeds TC by as much as possible. c. TC exceeds TR by...
-
Write a brief for a mobile app. Identify its purpose and target audience. Support your proposal with research evidence.
-
Discuss the functional significance of parallel versus serially arranged sarcomeres in terms of force output and velocity of shortening?
-
Chloe lifts a 6 6 - kg barbell from the ground to a height of 2 . 4 8 m. How much work was done on the barbell?
-
What is the maximum angular momentum L max that an electron with principal quantum number n = 5 can have? Express your answer in units of h .
-
Current Attempt in Progress Amy Dyken, controller at Crane Pharmaceutical Industries, a public company, is currently preparing the calculation for basic and diluted earnings per share and the related...
-
A company that uses job order costing incurred a monthly factory payroll of $203,000. Of this amount, $48,000 is indirect labor and $155,000 is direct labor. Prepare journal entries to record the (a)...
-
Evaluate the expression. (a) 10 C 3 (b) 10 P 3
-
In Problem use geometric formulas to find the unsigned area between the graph of y = f(x) and the x axis over the indicated interval. f(x) = x + 5; [0, 4]
-
How might you use 1k spectroscopy to distinguish between the following pairs of isomers? (a) CH 4 CH 2 OH and CHOCH 3 (b) Cyclohexane and 1-hexene (c) CH 3 CH 2 CO 2 H and HOCH 2 CH 2 CHO
-
The IR spectrum o phenyl acetylene is shown in figure. What absorption bands can youidentify? 100 80 60 40 -C=CH 20 4000 1500 Wavenumber (cm) 3500 3000 2500 2000 1000 500 Transmittance (%)
-
Where might the following compounds have IRabsorptions? (b) (c) .Co (a) C -H-CH2CH CH2OH
-
Annuity Payments Go to www.fcfcorp.com/onlinecalc.htm. Use the calculator to solve this problem. If you have $1,500,000 when you retire and want to withdraw an equal amount for the next 30 years, how...
-
Calculating Future Values Go to www.dinkytown.net and follow the Savings Calculator link. If you currently have $10,000 and invest this money at 9 percent, how much will you have in 30 years? Assume...
-
In 2023, Amanda and Jaxon Stuart have a daughter who is 1 year old. The Stuarts are full-time students and are both 23 years old. Their only sources of income are gains from stock they held for three...

Study smarter with the SolutionInn App


