In the following reaction, determine whether the alkyne has been oxidized, reduced, or neither. Using the answer
Question:
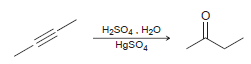
H2SO4. H20 HgSO4
Step by Step Answer:
One carbon atom is reduced from an oxidation state of 0 to a...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Sciences questions
-
Ammonia is burned to form nitric oxide in the following reaction: 4NH 3 + 5O 2 4N0 + 6H 2 0 (a) Calculate the ratio (lb-mole O 2 react/lb-mole NO formed). (b) If ammonia is fed to a continuous...
-
The initial steps of a laboratory synthesis of several prostaglandins reported by E. J. Corey (Section 7.15B) and co-workers in 1968 are outlined here. Supply each of the missing reagents: (a) (b)...
-
Two products are observed in the following reaction. (a) Suggest a mechanism to explain how these two products are formed. (b) Your mechanism for part (a) should be different from the usual mechanism...
-
Your buddy mentioned that she is thinking about suing under the qui tam law. You are not sure what that is. a. What is a qui tam provision? b. Can employees who do not work for the government sue...
-
Under what conditions would financial disintermediation increase? Under what conditions would disintermediation be advisable?
-
Delta Dawn's Bakery is considering purchasing a new van to deliver bread. The van will cost \(\$ 18,000\). Two-thirds \((\$ 12,000)\) of this cost will be borrowed. The loan is to be repaid with four...
-
Consider the regression model fit to the baseball data in Table B.22, using team ERA to predict the number of wins. a. Construct a normal probability plot of the residuals. Is there any indication of...
-
Calculating Real Rates of Return If Treasury bills are currently paying 8 percent and the inflation rate is 45 percent, what is the approximate real rate of interest the exact real rate?
-
Give two examples of errors which could occur when transposing information from source documents to journals. Explain what the flow-on effects of these could be if not detected immediately. Also...
-
Harbortown Marine Products (HMP) manufactures and sells various fixtures for boat cabins. One fixture uses a specialized fitting that is not used in any other HMP product. The management of HMP has...
-
Explain the general meaning of the following profit payoff matrix for oligopolists C and D. All profit figures are in thousands. a. Use the payoff matrix to explain the mutual interdependence that...
-
In January 2006, the yield on AAA-rated corporate bonds averaged approximately 5 percent; one year later, the yield on these same bonds had climbed to about 6 percent because the Federal Reserve...
-
a. Use the expansions of cos(2x + x) and cos(2x x) , to prove that: cos 3x + cos x + 2 cos 2x cos x b. Solve cos 3x + cos 2x + cos x > 0 for 0 < x < 360.
-
The employees of a transportation company complained to management that the men's washroom was very messy, despite regular cleaning. The employer implemented a log system that monitored the use and...
-
1. What do you think are the major reasons that more organizations are recruiting a diverse workforce? 2. What are some ways that recruiting could and should be done differently to attract more...
-
Think about the last time you had a problem with your own performance, either on the job or in one of your classes. Using the nine-step performance analysis model from Fournies, try to identify the...
-
If you were part of a team at a university that wanted to repeat the Scottish researchers study link between Alzheimer's and diet study described in , would you try to develop primary data on your...
-
What research options will work best for your business and industry? How can you ensure that these are the most helpful for your future success? Why? Make sure that you have clearly explained your...
-
Assume that a sample of bivariate data yields the correlation coefficient, r, indicated. Use Table 12.9 for the specified sample size and level of significance to determine whether a linear...
-
Which should drive action planning more, strengths or weaknesses? That is, is it more important to build on your strengths or to reduce your weaknesses? Explain.
-
A 1, 2-trans disubstituted cyclohexane must have either both groups axial or both groups equatorial. Explain.
-
Why is a 1, 3-cis disubstituted cyclohexane more stable than its transisomer?
-
Which is more stable, a 1, 4-trans disubstituted cyclohexane or its cis isomer?
-
Chang Company provided the following data for this year: Manufacturing overhead costs incurred: Indirect materials Indirect labor Property taxes, factory Utilities, factory Depreciation, factory...
-
ces On January 1, 2024, the Mason Manufacturing Company began construction of a building to be used as its office headquarters. The building was completed on September 30, 2025. Expenditures on the...
-
Primare Corporation provided the following data for last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor Manufacturing overhead applied...

Study smarter with the SolutionInn App


