N-Acetylazoles undergo hydrolysis more readily than regular amides. Suggest a reason for the enhanced reactivity of N-acetylazoles
Question:
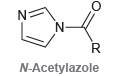
Transcribed Image Text:
N: 'N- N-Acetylazole
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
The lone pair of the nitrogen atom in this case is participa...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Pyrrole undergoes electrophilic aromatic substitution more readily than benzene, and mild reagents and conditions are sufficient. These reactions normally occur at the 2-osition rather than the...
-
A surface for which the electrostatic potential is negative delineates regions in a molecule that are subject to electrophilic attack. It can help you to rationalize the widely different chemistry of...
-
The explosion of an atomic bomb releases many radioactive isotopes, including strontium-90. Considering the location of strontium in the periodic table, suggest a reason for the fact that this...
-
What is unrealistic about the domino fault model?
-
What competitive advantage or disadvantage might an eBooks only textbook publisher have relative to McGraw-Hill, Pearson, and other large international publishers?
-
Suppose you are the manager of a new board shop (e.g., long boards, skateboards, kite boards, snowboards) located in your college or university community that caters primarily to the campus...
-
The tapered rod has a radius of r = (2 x/6) in. and is subjected to the distributed loading of w = (60 + 40x) lb/in. Determine the average normal stress at the center of the rod, B. w= (60+40x)...
-
Your manager is trying to determine what forecasting method to use. Based upon the fol-lowing historical data, calculate the following forecast and specify what procedure you would utilize. Month...
-
An auditor randomly samples 20 accounts receivable from among the 500 such accounts of a client's firm. The auditor lists the amount of each account and checks to see if the underlying documents...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
Propose a mechanism for the following transformation, and explain how you could use an isotopic labeling experiment to verify your proposed mechanism: [H,SO,] R.
-
Dimethylformamide (DMF) is a common solvent: (a) The 1 H NMR spectrum of DMF exhibits three signals. Upon treatment with excess LAH followed by water, DMF is converted into a new compound that...
-
Explain why a review of interim financial statements for a public company may provide a greater level of assurance than an SSARS review.
-
What are the similarities and differences between a general partnership and a limited partnership?
-
A corporation can behave in a socially responsible way by a. having strong policies in place to help the community. b. acting to reduce the negative impact a company has on the environment. c. being...
-
Amoral behavior is a. any illegal activity. b. not caring about the moral consequences of your action. c. when a person has a strong conviction behind their actions. d. a part of most religious...
-
In a journal article, Philipson and Jena (2006) study HIV survival and expenditures. Figure 13.9 shows trends in HIV survival and HIV expenditures in the US. a. Figure 13.9 (a) shows that HIV...
-
A business is in legal compliance if a. it follows all the laws of which it is aware. b. it cannot be proven guilty of violating any laws. c. it meets all local, state, and federal regulations. d. it...
-
In each scatter diagram, tell whether a linear or a quadratic model is appropriate for the data. If linear, tell whether the slope should be positive or negative. If quadratic, tell whether the...
-
Use integration by parts to evaluate the following. Check your answer by taking the derivative. x2e-xdx
-
Give structures for each of the following compounds. C8H10O: IR, 3150-3600 cm-1 (strong, broad); NMR, 1.17 (3/7, t, 7 = 8 Hz); 2.58 (2/7, q, / = 8 Hz); 6.0 (1/7, broad singlet, disappears with D20...
-
Give structures for each of the following compounds. C8H10O: IR, 3150-3600 cm-1 (strong, broad); NMR, 1.17 (3/7, t, 7 = 8 Hz); 2.58 (2/7, q, / = 8 Hz); 6.0 (1/7, broad singlet, disappears with D20...
-
How would you distinguish mesitylene (1, 3, 5-trimethylbenzene) from isopropylbenzene (cumene) by 13C NMR spectroscopy?
-
Choose from the following topics to create discussion: Humanity and the Natural Environment Housing and Home Furnishings Privacy and Socializing
-
On December 31, 2020, Pearl Company had $1,101,000 of short-term debt in the for January 21, 2021, the company issued 22,800 shares of its common stock for $42 per brokerage fees and other costs of...
-
During your interview process for the position of program manager for Booker, Tee, and How you are asked to explain the relationship between cost, schedule, and performance? what is your answer to...

Study smarter with the SolutionInn App


