Predict the expected number of signals in the 13 C NMR spectrum of each of the following
Question:
(a)
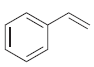
(b)
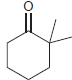
(c)
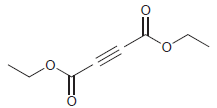
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a Six signals all of which appear in the region 100 150 ppm b ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many 1H NMR signals would you expect from each of the following compounds? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) OH CI
-
How many signals are produced by each of the following compounds in its a. 1H NMR spectrum? b. 13C NMR spectrum? 1. 2. 3. 4. 5. 6. CH3 OCHCH CH3 OCH2CH3 CI
-
For each of the following compounds, predict the number of signals and location of each signal in a 13 C NMR spectrum: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) H.
-
Explain why placing soy sauce in airtight bottles was more successful for long-distance shipping than simply placing the sauce in barrels.
-
What are three business applications of social networking sites?
-
Residents of neighboring towns in a state in the United States have an ongoing disagreement over who lays claim to the higher average price of a single-family home. Since you live in one of these...
-
In a recent study on risk factors for strokes, the American Heart Association (AHA) collected data on age, systolic blood pressure, diastolic blood pressure, and risk of stroke; risk is interpreted...
-
Jabber Foods produces specialty soup sold in jars. The projected sales in dollars and jars for each quarter of the upcoming year are as follows: Jabber anticipates selling 220,000 jars with total...
-
Miller Company manufactures a line of lightweight running shoes. CEO Richard Miller estimated that the company would incur $7,000,000 in manufacturing overhead during the coming year. Additionally,...
-
Mr B aged 52 years, has earned rupees 75,00,000 out of his business. His ex-wife gifted him a car worth rupees 8 lakh. He spent a total of rupees 20 lakh during a family trip. He won a lottery of 16...
-
Rank the signals of the following compound in terms of increasing chemical shift. Identify the proton(s) giving rise to each signal: CI. .
-
When 1-methylcyclohexene is treated with HCl, a Markovnikov addition is observed. How would you use 1 H NMR spectroscopy to determine that the major product is indeed the Markovnikov product?
-
The last paragraph establishes all of the following about The Tin EXCEPT: F. The Tin is over twelve hundred years old. G. Once a literary work is dead, it ceases to have any cultural significance. H....
-
Genesis Inc. was formed in 2015. Its current \(\mathrm{E} \& \mathrm{P}\) and distributions to shareholders made each year are listed below. What is Genesis Inc.'s year-end accumulated \(E \& P\)...
-
The boiling point for species A at 1 bar is reported to be 250 K, and you desire to know the saturation pressure at 300 K. Knowing the enthalpy of vaporization, you apply the Clausius-Clapeyron...
-
Beatles Inc. has 1,000 shares issued and outstanding. The shares are owned as follows: How many shares is Paul consider to own for redemption purposes? Name Paul John Corp. (Paul owns 70% of John...
-
At room temperature, iron exists in the ferrite phase (-Fe). At 912C, it goes through a phase transformation to the austenite phase (-Fe). Which phase of iron has stronger bonds? Explain.
-
Develop an approach to construct threshold curves for the business customers.
-
Susan is a single taxpayer, 26 years of age, with AGI of $28,000 and no tax-exempt income. She did not have minimum essential coverage for 8 months in 2018. Compute Susans individual shared...
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
1, 6-Hexanediamine, a starting material needed for making nylon, can be made from 1, 3-hutadiene. How would you accomplish thissynthesis? H2NCH2CH2CH2CH2CH2CH2NH2 H2C=CHCH=CH2
-
A chemist in need of 2, 2-dimethylpentanoic acid decided to synthesize some by reaction of 2chloro-2-methylpen Lane with NaCN, followed by hydrolysis of the product. After the reaction sequence was...
-
Show how you might prepare the anti-inflammatory agent ibuprofen starting from isobutyl benzene. More than one step isneeded. CO2H Isobutylbenzene Ibuprofen
-
If the nominal interest rate is 14%, and inflation is 4%, what is the real interest rate? 22.A bond investment yielded 8%. If inflation was 3%, what real return did the bond offer? 23.The following...
-
Bierce Corporation has two manufacturing departments--Machining and Finishing. The company used the following data at the beginning of the year to calculate predetermined overhead rates: Estimated...
-
Discuss how communication channels have changed over the years due to technology.
A New Silver Solution A Quantum Leap In Silver Technology 5th Edition - ISBN: 1887938435 - Free Book

Study smarter with the SolutionInn App


