Predict the major product of the following reactions. a. b. c. d. NBS Heat or light Na,Cr,0,
Question:
a.
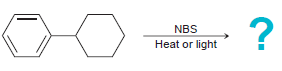
b.
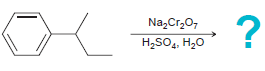
c.
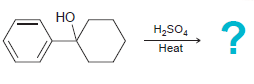
d.
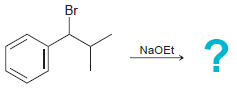
NBS Heat or light Na,Cr,0, H,SO,, H20
Step by Step Answer:
a b c...View the full answer



Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Sciences questions
-
Predict the product of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) HBr (excess), OH (1) NaH OH Br2 (excess) H20 H3C OH excess QH (1) NaH,Br (2) heat HNO3, H2SO4 NaNH2, NHa (1) NaOH...
-
Predict the major products of the following reactions: (a) (b) (c) cat. H2SO H,O (2) H202. NaOH (1) Hg(OAc), H2O/THF (2) NaBH4, NaOH
-
Predict the products of the following reactions: (a) (b) (c) (d) (e) (f) CI )LiAIH (2)H30 CI LiAIHO--Bu) (1) DIBALH OCH (2) H20 (I) excess CH2Mgl CI ) Ho (1) DIBAL-H (-78C) (2) H,0
-
What constant should replace the question mark in this system so that the solution set is {(1, 1, 1)}? 2x - 3y + -5x + 2y z = 0 - 2 = x + y + 2z = ? 4 -4
-
View the YouTube video: https://www.youtube.com/watch?v=PHJ8eybXJdw Answer the Question, How to Manage Team Conflict. Briefly describe a team conflict you observed or were involved in that had...
-
Show that for the Lagrangian density (16.7) of a complex scalar field, the field equation (16.14) reduces to the two Klein-Gordon equations given by Eq. (16.8). Data from Eq. 16.7 Data from Eq. 16.8...
-
During the assembly of an exhaust valve, sufficient distance must be maintained between the valve tip and the cylinder wall. If \(85 \%\) of valve assemblies have the required distance, use Table 1...
-
The model for making tactical decisions described in the text has six steps. These steps are listed, out of order, below. Required: Put the steps in the correct order, starting with the step that...
-
4-0 stainless steel sutures used for orthopedic applications are subjected to a heat treating process. The sutures are drawn through an oven at a rate of 0.5 meters per minute. The oven temperature...
-
Bill has just completed his exams for the academic year and wants to celebrate by seeing every movie showing in theaters in his town and in six other neighboring cities. If he travels to another...
-
The following two drawings are resonance structures of one compound: But the following two drawings are not resonance structures: They are, in fact, two different compounds. Explain. Not resonance...
-
A compound with molecular formula C 8 H 10 produces an IR spectrum with many signals, including 3108, 3066, 3050, 3018, and 1608 cm -1 . The 1 H NMR spectrum of this compound exhibits a singlet at...
-
(a) Show that if I2 = 10.0 I1, then (2 = (1 + 10.0 dB. (A factor of 10 increase in intensity corresponds to a 10.0 dB increase in intensity level.) (b) Show that if I2 = 2.0 I1, then (2 = (1 + 3.0...
-
With reference to CAATs, explain briefly: (i) The test data technique. (ii) Three types of audit software.
-
Frequently, the project's tasks are not well defined, and there is an urge to "get on with the work," since time is critical. How serious is it to minimize the planning effort and get on with the...
-
How does the reliability of healthcare services affect the quality of care you receive? What type of healthcare service do you find to be the least reliable in delivering a quality product? What type...
-
A hospital made 225 medication errors last year. Of these errors, 30 percent were errors with the prescription, while 70 percent were errors made while dispensing the medication. The hospital...
-
Review Figure A-4, plot the points on coordinate axes, and then state whether each of the following paired observations is on, above, or below the x axis and on, to the left of, or to the right of...
-
Using the information given in Section 11.5 and the mass energy conversion equation, compute the binding energy (in MeV) and the binding energy per nucleon for hydrogen-2 (deuterium).
-
SCHEDULE OF COST OF GOODS MANUFACTURED The following information is supplied for Sanchez Welding and Manufacturing Company. Prepare a schedule of cost of goods manufactured for the year ended...
-
Give the missing reactant or product in each of the following equations. (a) (b) + 0 Br HBr (only product)
-
Outline a laboratory preparation of each of the following compounds. Each should be prepared from an alkene with the same number of carbon atoms and any other reagents. The reactions and starting...
-
Deuterium (D, or 2H; is an isotope of hydrogen with atomic mass - 2. Deuterium can be introduced into organic compounds by using reagents in which hydrogen has been replaced by deuterium. Outline...
-
What theorists are most likely to argue the city's form and growth come from decisions made by people and organizations that control wealth and other key resources?
-
Explain the difference between glucogenic and ketogenic amino acids
-
Describe what is an arrangement where workers who don't join a union must make payments equal to union dues and fees to get union representation services?

Study smarter with the SolutionInn App


