Predict the major product(s) for each of the following reactions: 1) Hg(OAC)2, -0 2) NABH, ? -
Question:
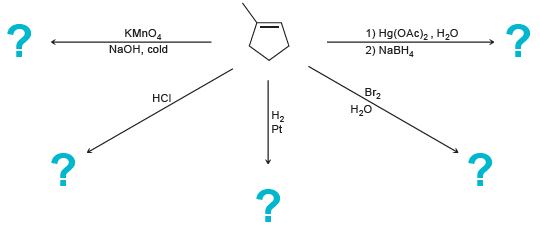
Transcribed Image Text:
1) Hg(OAC)2, Н-0 2) NABH, ? - КМпO, NaOH, cold Br2 НС H20 Pt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (17 reviews)
HO OH En KMnO4 ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) HO OH NH2 HA (cat.) (1) HCN (2) LiAIH4 (3) H2o mCPBA
-
Data obtained from asking the wrong questions at the wrong time or in the wrong place can lead to misleading summary statistics. Explain why the following collection procedures are likely to produce...
-
Ensuring inmate and staff safety is of the utmost importance in a correctional facility. Write an essay in which you will focus on some of the possible techniques that prisons use. Why is inmate...
-
A city government is considering building a new system of lighted bike paths. A council or supporting their construction lists the following as potential benefits of the paths: (1) more enjoyable...
-
Divide the methanol reaction operation in Example 6.6 into five consecutive stages in series. Feed the \(\mathrm{CO}\) reactant entirely into the first operation at \(25^{\circ} \mathrm{C}\) and \(1...
-
Blanche Creek has engaged your firm because she has been charged with failure to file her 2013 federal Form 1040. Blanche maintains that the reasonable cause exception should apply. During the entire...
-
Financial data for Joel de Paris, Incorporated, for last year follow: Joel de Paris, Incorporated Assets Cash Accounts receivable Inventory Plant and equipment, net Balance Sheet Beginning Balance...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
For each pair of compounds below, determine whether they are identical compounds, constitutional isomers, stereoisomers, or different conformations of the same compound: (a) (b) (c) (d) (e) (f) (g)...
-
Draw a Lewis structure for each of the following compounds: (a) C 2 H 6 (b) C 2 H 4 (c) C 2 H 2 (d) C 3 H 8 (e) C 3 H 6 (f) CH 3 OH
-
Verify that the price of the 12% interest rate cap in Figure 25.6 is $3.909. In Figure 25.6 Year YYear 2 Year 3 $6.689 $6.799 $1.078 $3.184 $0.00 $1.463 $0.00 $0.250 $0.00 $0.00
-
Again, you are dealt 4 cards. The first card is a spade, the second a heart, the third a diamond, and the fourth a club. What is the probability that you are dealt all 4 aces?
-
An advertising executive decides that a television commercial should be shown on two television stations. If three television stations serve the area the company wants to reach, how many possible...
-
Your investment advisor has a portfolio of 75 stocks: 40 high-growth stocks and 35 high-dividend stocks. Of the 40 high-growth stocks, 25 have increased in value over the last year, whereas 10 of the...
-
The Tastee Coffee Company is about to begin selling a new gourmet coffee. Company managers know that the probability the product will be profitable is 20 %. They also know that the probability that...
-
You have drawn three diamonds, one spade, and one heart from a deck of cards. If you discard the spade and the heart, what is the probability of your drawing two cards from the remaining 47 cards to...
-
Use a calculator or program to compute the first 10 iterations of Newtons method when it is applied to the following functions with the given initial approximation. Make a table similar to that in...
-
7 A 29-year-old, previously healthy man suddenly collapses at a party where legal and illicit drugs are being used. Enroute to the hospital, he requires resuscitation with defibrillation to establish...
-
Show the elimination products of thesereactions: Br ELOH a) CH,CH,CHCH,CH, + CH;CH20 ,, CI CI . EIOH + OH b) + CH,CH20 EIOH
-
Show the products, including stereo chemistry, of these eliminationreactions. CH;CH2 Br Br Br b) H C-CH + CH,CH20 Ph ELOH Ph C-C EIO H+ OH Ph H2O Ph
-
What product would be expected from the elimination reaction of (1R, 2S)-1-bromo-1, 2-dipheylpropane using sodium ethoxide in ethanol as the solvent?
-
ABC Hospital is one of two hospitals in the community. The Hospital offers all major specialty services (e.g. cardiology, neurology, pulmonology, etc.) The Hospital is approached by a cardiologist...
-
You obtained job-related information about the HR assistant position from O*Net. Answer the following sections based on that information. QUESTIONS: The following is the list of questions included in...
-
On January 8, the end of the first weekly pay period of the year, Regis Company's employees earned $27,760 of office salaries and $70,840 of sales salaries. Withholdings from the employees' salaries...

Study smarter with the SolutionInn App


