Predict the product(s) for each of the following reactions: (a) (b) (c) (d) Br Cl2 AICI, HNO3
Question:
(a)
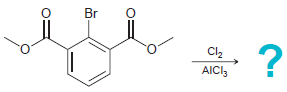
(b)
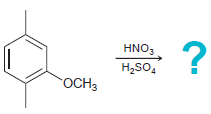
(c)
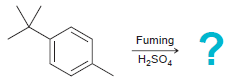
(d)
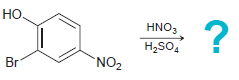
Transcribed Image Text:
Br Cl2 AICI, HNO3 H,SO, OCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a ...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The enthalpy change for each of the following reactions was calculated using bond energies. The bond energies of XO, YO, and ZO are all equal. XX + O=O XOOX; H = 275 kJ YY + O=O YOOY; H = +275 kJ...
-
The reaction: 4A + 3B 1 2C + D is studied. Unknown masses of the reactants were mixed. After a reaction time of 1 hour the analysis of the mixture showed 2 kmol of A, 1 kmol of B and 4 kmol of C....
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) (b) (c) (d) ...
-
Find each product. (8 m)( + m)
-
What is Evidence Informed Practice? What is Delaware's Mandatory reporting law for child abuse and neglect? What kinds of cases would you expect to find in civil proceedings? What is the role of a...
-
It is possible to adapt some of the tools used in linear regression to certain nonlinear cases. Suppose, for example, you wanted to link independent variable x to dependent variable y using the...
-
The gas mixture in a helium-neon laser (end mirrors removed) emits light at \(633 \mathrm{~nm}\) with a Doppler-broadened spectral width of about \(1.5 \times 10^{9} \mathrm{~Hz}\). Calculate the...
-
1. How did the U.S. Strategic Command adjust to unexpected issues as it implemented extreme programming? 2. What types of changes accompany the extreme programming methodology? 3. Why could a...
-
It is evident that Central Banks in advanced capitalist nations such as Australia are serious about reducing annual CPI inflation, which was 5.4 per cent in the September 2023 quarter. Raising the...
-
Matt and Peter are the employees of a reputed company. The company management decided to increase the work space in the office, so the management introduced open office system. There are no...
-
Propose a plausible synthesis for each of the following transformations: (a) (b) (c) (d) H OCH3 NO2 Br OCH3 OCH3 Br NO2
-
Each of the following compounds can be made with a Friedel-Crafts acylation. Identify the acyl chloride and the aromatic compound you would use to produce each compound. (a) (b) O2N OCH3 H
-
Carver Department Stores, Inc., constructs its own stores. In the past, no cost has been added to the asset value for interest on funds borrowed for construction. Management has decided to correct...
-
Which bond usually will have a higher liquidity premium: one issued by a large company or one issued by a small company?
-
Why have prepackaged bankruptcies become so popular in recent years?
-
Why are rates of return superior to dollar returns when comparing different potential investments?
-
Indicate whether each of the following statements is true or false by writing T or F in the answer c olumn. An inter v ivos trust is created by a will.
-
Consider the following information for a fish farm that has an infinite demand for fish produced at 5 per kg. a. Fill in all the blanks in the table. b. Verify that MRP L for the fish farm can be...
-
If our model of rent control allowed for unrestricted subletting, who would end up getting apartments in the inner circle? Would the outcome be Pareto efficient?
-
The Pletcher Transportation Company uses a responsibility reporting system to measure the performance of its three investment centers: Planes, Taxis, and Limos. Segment performance is measured using...
-
Ln view of the bromonium-ion mechanism for bromine addition, which of the products in your answer to Problem 7.25 (p.305) are likely to be the major ones?
-
Contrast the results in problem 7.28 with those o be expected when cis- and trans-a (not isotopical substituted) are subjected to the same reaction Cmditions.
-
Using the information in the previous problem and in Eq. 7.7, contrast the relative amounts of axial conformations in samples of methylcyclohexane and tert-butylcyclohexane.
-
Last year, a city had three school districts: North with a population of 5200 children, South with a population of 10600 children, West, with a population of 15100 children. Use Hamilton, Adams,...
-
Prepare the journal entry to record pension expense and the employer ? s contribution to the pension plan in 2 0 2 0 . ( Credit account titles are automatically indented when amount is entered. Do...
-
Concord Department Store uses a perpetual inventory system. Data for product E2-D2 include the following purchases. Date Number of Units Unit Cost May 7 July 28 45 30 $13 15 On June 1, Concord sold...

Study smarter with the SolutionInn App


