Predict the products for each of the following transformations. a. b. aso heat 1) TsCl, py -OH
Question:
a.
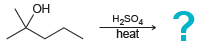
b.
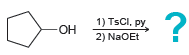
Transcribed Image Text:
ОН Нaso heat 1) TsCl, py -OH 2) NaOEt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (17 reviews)
a b O...View the full answer

Answered By

Omar ELmoursi
I'm Omar, I have Bachelor degree in Business and Finance, My unique approach is to help students with questions and assignments, I can teach Business, Math, Accounting, Managerial Accounting, Economy, Human resources management, organizational behavior, project management, I have experience dealing with different types of students and teach them how to deal with different types of exercises.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products for each of the following: a. b. c. d. e. f. 1) O3 2) DMS 3) Excess LAH 4) H20 1) O3 2) DMS 3) Excess LAH 4) H20
-
Predict the products for each of the following. a. b. c. d. e. f. g. h. 1) RCO,H 2) MeMgBr 3) , 1) Hg(OAc)2, MeOH 2) NaBH,
-
Predict the products for each of the following reactions: H2 Lindlar's catalyst 2 Pt
-
If f (x, y, z) = 2xyz - 1 + x2z3, find the following. (a) f/x (b) f/y (c) f/z
-
"We're in the furniture business, and I'll bet you have used some of our stuff if you spent any time in your college dorms. But, I'll tell you something, there's not much profit in that business- too...
-
Consider a system with a total of 150 units of memory, allocated to three processes as shown: Apply the banker's algorithm to determine whether it would be safe to grant each of the following...
-
In which engine values are used? (a) Two-stroke engine (b) Four-stroke engine (c) In both engines (d) None of the above
-
Bunyan Lumber, LLC, harvests timber and delivers logs to timber mills for sale. The company was founded 70 years ago by Pete Bunyan. The current CEO is Paula Bunyan, the granddaughter of the founder....
-
How do distillation systems incorporate heat integration and energy recovery techniques to optimize energy consumption, and what are the latest advancements in these areas ?
-
Recognition accuracy that measures the percentage of orders that are taken correctly is 86.8%. Suppose that you place order with the bot and two friends of yours independently place orders with the...
-
Compound Y has molecular formula C 7 H 12 . Hydrogenation of compound Y produces methylcyclohexane. Treatment of compound Y with HBr in the presence of peroxides produces the following compound:...
-
Distinguish between beta (or market) risk, within-firm (or corporate) risk, and stand-alone risk for a project being considered for inclusion in the capital budget. Which type of risk do you believe...
-
List the legal elements of fraud.
-
Why does business risk vary from industry to industry?
-
What happens to the component costs of debt and equity when the debt ratio is increased? Why does this occur?
-
How does operating leverage affect business risk?
-
Using the Hamada equation, explain the effects of financial leverage on beta.
-
If a firm fails to consider growth options, would this cause it to underestimate or overestimate projects NPVs? Explain.
-
In 2018, Michael has net short-term capital losses of $1,500, a net long-term capital loss of $27,000, and other ordinary taxable income of $45,000. a. Calculate the amount of Michaels deduction for...
-
What are the risks and liability factors in an audit? What are the implications to the auditor? What are the implications to the organization? How can the auditor mitigate these risks and liability...
-
2-Chloro-2-mcthylpropane reacts with water in three steps to yield 2-methyl- 2-propanol. The first step is slower than the second, which in turn is much slower than the third. The reaction takes...
-
An Add curved arrows to the mechanism shown in Problem 5.34 to indicate the electron movement in each step.
-
The reaction of hydroxide ion with chloromethane to yield methanol and chloride ion is an example of a general reaction type called a nucleophilic sub- Situation reaction: HO- + CH3C1 CH3OH + C1-...
-
Write an equation for the polynomial graphed below 5+ 4 + -5-4-3-2 y = 3. 2- 1 + + 2 3 4 5 -2 345 -5+
-
How can ethical leadership principles be leveraged to navigate the complexities of organizational dynamics and promote a culture of integrity, accountability, and social responsibility ?
-
Consider the 12-QAM constellation shown in Figure 2 and the baseband pulse, g(t) depicted in Figure 3. The 12 passband transmit waveforms are represented as Sm1(t) = Amig(t) cos (2fet) - Amag(t) sin...

Study smarter with the SolutionInn App


