Predict which of the following two compounds will undergo an E2 reaction more rapidly: CI
Question:
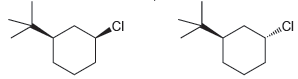
Transcribed Image Text:
CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (19 reviews)
Because of the bulky tertbutyl group the first compou...View the full answer

Answered By

Sandhya Sharma
I hold M.Sc and M.Phil degrees in mathematics from CCS University, India and also have a MS degree in information management from Asian institute of technology, Bangkok, Thailand. I have worked at a international school in Bangkok as a IT teacher. Presently, I am working from home as a online Math/Statistics tutor. I have more than 10 years of online tutoring experience. My students have always excelled in their studies.
4.90+
119+ Reviews
214+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would a structural effect that destabilizes the acid component of a conjugate acid-base pair affect its acidity? Use your analysis to predict which of the following two compounds is more basic....
-
Which of the following two compounds would eliminate HBr more rapidly in basic solutions? CH2Br or CH2Br
-
(a) Two stereoisomers of a bromodecalin are shown. Although the difference between these stereoisomers may seem trivial, one isomer undergoes elimination with KOH much faster than the other. Predict...
-
Elaborate the basic functioning of a hydraulic lift. Include the terms pressure, density, and buoyant force in the explanation. Give one example of a hydraulic lift you have encountered.
-
A natural deposit of soil is found to have a water content of 20% and to be 90% saturated. What is the void ratio of this soil?
-
Given the analog transfer function \[H(s)=\frac{1}{\left(s^{2}+0.76722 s+1.33863 ight)(s+0.76722)},\] design transfer functions corresponding to discrete-time filters using both the impulseinvariance...
-
Consider a market with two securities, one of them risk free with rate of return \(R\), and three future states. Suppose that the risky security has a present value \(S_{1}\) and can take three...
-
Future Value and Multiple Cash Flows Paradise, Inc., has identified an investment project with the following cash flows. If the discount rate is 8 percent, what is the future value of these cash...
-
How much would a business have to invest in a fund to receive $21,000 at the end of every month for 7 years? The fund has an interest rate of 4.50% compounded monthly and the first withdrawal is to...
-
A circular hoop is suspended in a horizontal plane by three strings, each of length l, which are attached symmetrically to the hoop and are connected to fixed points lying in a plane above the hoop....
-
The Organization for African Unity has vigorously opposed the export of hazardous waste from industrialized countries to developing nations. They call this practice toxic terrorism. Explain.
-
Compare integrated pest management to integrated waste management. How does each reduce potential damage to the environment?
-
Can diversity be a strategic advantage to organizations?
-
Picnic Time produces a picnic basket that is sold for $ 1 0 0 per unit. Assume the company produced and sold 4 , 0 0 0 baskets during July. There were no beginning or ending inventories. Variable and...
-
Hudson Lighting sells high end light fixtures. The company reported the following contribution margin income statement for 2 0 2 2 . Hudson Lighting Contribution Income Statement For Year Ended...
-
On January 2, Ramirez purchased equipment costing $23,500. The equipment has an estimated salvage value of $1,480 and an estimated useful life of years. Required: Compute the depreciation expense for...
-
Oriole Corporation received cash of $36900 on September 1, 2025 for one year's rent in advance and recorded the transaction with a credit to Unearned Rent Revenue. What is the December 31, 2025...
-
A facility manager regularly visits staff on the job and expresses concern for helping everyone accomplish their work. It seems to have a positive motivating effect on work output. Which management...
-
Why is it sometimes difficult to decide whether an observed change is physical or chemical?
-
Find the image of x = k = const under w = 1/z. Use formulas similar to those in Example 1. y| y = 0 -21 -2 -1 -1, /1 12 T -1 -1 y= -2 x =0
-
Predict the products from reaction of 1-hexyne with the following reagents: (a) 1 equiv HBr (b) 1 equiv Cl2 (c) H2, Lindlar catalyst (d) NaNH2 in NH3, then CH3Br (e) H2O, H2SO4, HgSO4 (f) 2 equiv HC1
-
Predict the products from reaction of 5-decyne with the following reagents: (a) H2 Lindlar catalyst (b) Li in NH3 (c) 1 equiv Br2 (d) BH3 in THF, then H2O2, OH (c) H2O, H2SO4, HgSO4 (f) Excess H2,...
-
Predict the products from reaction of 2-hexyne with the following reagents: (a) 2 equiv Br2 (b) 1 equiv HBr (c) Excess HBr (d) Li in NH3 (e) H2O, H2SO4, HgSO4
-
Choose from the following topics to create discussion: Humanity and the Natural Environment Housing and Home Furnishings Privacy and Socializing
-
On December 31, 2020, Pearl Company had $1,101,000 of short-term debt in the for January 21, 2021, the company issued 22,800 shares of its common stock for $42 per brokerage fees and other costs of...
-
During your interview process for the position of program manager for Booker, Tee, and How you are asked to explain the relationship between cost, schedule, and performance? what is your answer to...

Study smarter with the SolutionInn App


