Provide a systematic name for each of the following compounds. a. b. c. d. e. H. CH3
Question:
a.
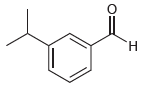
b.
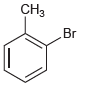
c.
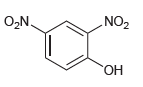
d.
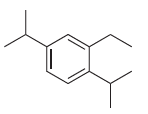
e.
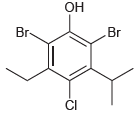
Transcribed Image Text:
H. CH3 Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
a 3isopropylbenzaldehyde or metaisop...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Provide a systematic name for each of the following compounds below: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) (q)
-
Give the systematic name for each of the following compounds a. b. (CH3)3CCH2CH2CH2CH(CH3)2 C. d. (CH3CH2)4C e. CH2CH2CH2CH2CH2NHCH2CH3 f. g. h. i. j. k. CH3OCH2CH2CH2OCH3 Br CH3HCH2CH2CHCH2CH2CH3 CH...
-
Provide a systematic name for each of the following compounds: (a) (b) (c) (d)
-
Define pricing practices of tesla INC as well as market structure.
-
The laboratory test results on a sand are as follows: e max = 0.91, e min = 0.48, and Gs = 2.67. What would be the dry and moist unit weights of this sand, in lb/ft3, when densified at a moisture...
-
William Leiderman is a portfolio manager for a US pension funds domestic equity portfolio. The portfolio is exempt from taxes, so any differences in the taxation of dividends and capital gains are...
-
Green Spaces Make Kids Smarter A recent article claims that "Green Spaces Make Kids Smarter." The study described in the article involved 2,623 schoolchildren in Barcelona. The researchers measured...
-
Nonfinancial measures of quality, manufacturing cycle efficiency. (CMA, adapted) Torrance Manufacturing evaluates the performance of its production managers based on a variety of factors, including...
-
Bramble Corp, applies overhead on the basis of machine hours. Given the following data, what is the amount of overhead applied and the amount by which it is under- or overapplied for the period?...
-
A coal gas is to be freed of benzene by scrubbing with wash oil as an adsorbent and the light oil recovered by stripping the resulting solution with steam. The circumstances are: Absorber . Gas inlet...
-
Compare the structures of 1,4-pentadiene and divinyl amine: The first compound does not absorb UV light in the region between 200 and 400 nm. The second compound does absorb light above 200 nm. Using...
-
Provide a systematic name for each of the following compounds. a. b. c. d. e. H.
-
Although 2, 2-dimethylhexane has a molecular weight of 114, no peak is observed at m/z = 114. The base peak in the mass spectrum occurs at M57. a. Draw the fragmentation responsible for formation of...
-
Explain how High-Frequency Traders (HFT) negatively affect market liquidity? Explain in detail so that it is easy to understand.
-
List five 5 fraud risks banks are exposed to, and give measures to meet such risks.
-
Between watching a financial news network on cable, reading articles in some business magazines, and listening to a co-worker recount his story of doubling his portfolio in six months, Brad is now...
-
Discuss two specific financial services provided by banks to meet the needs of small and medium enterprise (SME)
-
Find the price for the bond. Par Value: 5,000 Coupon Rate: 8% Years to Maturity: 15 Yield to Maturity: 11% Price: ?
-
Determine the monthly payment for the installment loan. Unless told otherwise by your instructor, you may either use Table 10.2 or the installment payment formula. Table 10.2: Number Annual...
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
Which of the following structures are identical? (Yellow-green =Cl) (a) (b) (c) (d)
-
A Assign R or S configuration to the chirality centers in the following molecules (blue =N): (b) (a) Adrenaline Serine
-
Which, if any, of the following structures represent meso compounds? (Blue = N, yellow-green =Cl.) (b) (c)
-
Consider the vectors u=2ij2k and v=2i+2j+4k. Find the vector components of u that are parallel and perpendicular to v.
-
1. Determine whether the following sets form subspaces and justify your answers. (a) {(1, 2): 122 = 0} in R (b) {(1, 2) |1|-|x2|} in R2 (c) {(x1, x2, x3): x3 = 21 or x3 = 12} in R3 (d) {(x1, x2, x3):...
-
Solve the system of two equations for y two equations are: A1=ax+by A2=cx+dy

Study smarter with the SolutionInn App


