Rank this group of compounds in order of increasing boiling point. -NH2 -N-
Question:
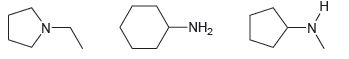
Transcribed Image Text:
н -NH2 -N-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
N Incr...View the full answer

Answered By

Pranav Sikerwar
Axis Techsoft Solutions
Content Developer, 2011-12
Developed ppt and flash files of educational content of Maths and Physics for IIT Alumni Startup.
Academic Manager,
Learnix Edutech, 2012-17
Developed collaborations with Various schools in providing e-solutions related to Maths, Physics, Aptitude and analytical skills.
Advanced Maths Faculty,
Study Vault, DPS Indirapuram
Taught Maths for competition level for students of 9th to 12th.
-IIT-JEE advanced level for Class 11th-12th
-Olympiad/NTSE/KVPY preparation for Class 9th-10th
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank each group of compounds in order of increasing boiling point, (a) Cyclohexane, cyclohexanol, chlorocyclohexane; (b) 2,3-dimethyl-2-pentanol, 2-methyl-2-hexanol, 2-heptanol.
-
Rank each set of compounds in order of increasing boiling points. (a) Triethylamine, di-n-propylamine, n-propyl ether (b) Ethanol, dimethylamine, dimethyl ether (c) Diethylamine, diisopropylamine,...
-
Arrange each group of compounds in order of increasing basicity. (a) CH3COO-, ClCH2COO-, PhO- (b) Sodium acetylide, sodium amide, and sodium acetate (c) Sodium benzoate, sodium ethoxide, and sodium...
-
4. In cells B4:B15, type a yearly salary for each staff member. Format these values as Currency with no decimal places. 5. Calculate the new salary with a 10% increase in column C. 6. Calculate the...
-
A company's sales in 2007 were $250,000 and in 2008 were $287,500. Using 2007 as the base year, the sales trend percent for 2008 is: a. 87% b. 100% c. 115% d. 15% e. 13%
-
You are serving on a jury. A plaintiff is suing the city for injuries sustained after a freak street sweeper accident. In the trial, doctors testified that it will be five years before the plaintiff...
-
What is the short-run impact of immigration on the wage of native workers? What is the long-run impact?
-
The pull strength of a wire bond is an important characteristic. The following table gives information on pull strength (y), die height (x1), post height (x2), loop height (x3), wire length (x4),...
-
Calculate the details of working capital? current ratio = 3 . 5 liquid ratio = 2 . 5 stock turnover ratio = 6 time debtor collection period = 3 months gross profit ratio = 2 0 % net working capital...
-
If v(t) = 160 cos 50t V and i(t) = -20 sin(50t - 30) A, calculate the instantaneous power and the average power.
-
Draw all constitutional isomers with molecular formula C 3 H 9 N, and provide a name for each isomer.
-
Identify whether each of the following compounds is expected to be water soluble: (a) (b) (c) -NH2 -NH2
-
Failure to make an adjusting entry to recognize accrued salaries payable would cause which of the following? (Hint: See Tip for how to analyze the effects.) a. An understatement of expenses,...
-
A proton moves in a region of uniform magnetic field, as shown in Figure Q24.23. The velocity at one instant is shown. Will the subsequent motion be a clockwise or counterclockwise orbit? FIGURE...
-
At a particular moment, the current in the primary coil is clockwise, as viewed from the secondary coil. At the center of the secondary coil, the field from the primary coil is A. To the right. B. To...
-
Identify the element for each of these electron configurations. Then determine whether this configuration is the ground state or an excited state. a. \(1 s^{2} 2 s^{2} 2 p^{5}\) b. \(1 s^{2} 2 s^{2}...
-
What is the ground-state electron configuration of calcium \((Z=20)\) ? A. \(1 s^{2} 2 s^{2} 2 p^{6} 3 s^{2} 3 p^{8}\) B. \(1 s^{2} 2 s^{2} 2 p^{6} 3 s^{2} 3 p^{6} 4 s^{1} 4 p^{1}\) C. \(1 s^{2} 2...
-
An electron collides with an atom in its ground state. The atom then emits a photon of energy \(E_{\text {photon }}\). In this process the change \(\Delta E_{\text {elec }}\) in the electron's energy...
-
1. Why is the millennial generation, in particular, so important to Amex? 2. This case focuses primarily on the battle between Chase and Amex over millennial consumers. But, as the content in this...
-
Citing a scientific article, explain in your own words, how DNA fingerprinting has been used in forensic science to solve crimes and why it may not always be accurate or effective.
-
When DNA is treated with 0.5 M NaOH at 25C, no reaction takes place, but when RNA is subjected to the same conditions, it is rapidly cleaved into mononucleotide 2- and 3-phosphates. Explain.
-
Indicate whether you would expect imidazole to have high or low water solubility, and why.
-
What product is expected when 4-(dimethylamino)pyridine reacts with CH3I?
-
"Managing Away Bad Habits Team Assignment Organizational Behavior IILeadership Assigned is ashort case from the exercise Managing Away Bad Habits. The task is to develop a turnaround strategy for...
-
"Managing Away Bad Habits Team Assignment Organizational Behavior IILeadership Assigned is ashort case from the exercise Managing Away Bad Habits. The task is to develop a turnaround strategy for...
-
11 The APRN unit director is working at an agency that has a high incidence of medication errors, specifically with heparin. One recent error had a patient receive twice the prescribed dose. Which...

Study smarter with the SolutionInn App


