(R)-Limonene is found in many citrus fruits, including oranges and lemons: Draw the structures and identify the...
Question:
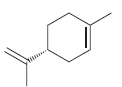
Draw the structures and identify the relationship of the two products obtained when (R)-limonene is treated with excess hydrogen in the presence of a catalyst.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
exces...View the full answer

Answered By

PU Student
cost accounting
financial accounting
auditing
internal control
business analyst
tax
i have 3 years experience in field of management & auditing in different multinational firms. i also have 16 months experience as an accountant in different international firms. secondary school certification.
higher secondary school certification.
bachelors in mathematics.
cost & management accountant
4.80+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(R)-Limonene is found in many citrus fruits, including oranges and lemons: For each of the following compounds identify whether it is (R)-limonene or its enantiomer, (S)-limonene: a. b. c. d.
-
Tartaric acid is found in many fruits, including grapes, and is partially responsible for the dry texture of certain wines. Calculate the pH and the tartarate ion (C4H4O62-) concentration for a 0.250...
-
Citric acid (C6H8O7) is the acid found in citrus fruits. It can lose a maximum of three H+ ions in the presence of a base. Write the chemical equations for citric acid acting stepwise as a...
-
Find steady state current i of a RL circuit with L=2 H and R=892 which can be modeled by the following differential equation: 2di/dt+8i=v(t) where v(t) = 16 cos(10t -45) with phasor technique. Also...
-
a. To what extent can we really see the world as it really is? b. To what extent is truth a function of power? c. To what extent to texts have stable meanings? d. What are the best Christian...
-
J. Hill has the following assets and liabilities on 30 November 2016: Accounts payable 2,800; Equipment 6,200; Car 7,300; Inventory 8,100; Accounts receivable 4,050; Cash at bank 9,100; Cash in hand...
-
Suppose that the recycle times of two cameras are recorded. The extension of the positive real line \(R\) is to take the sample space to be the positive quadrant of the plane \[ S=R^{+} \times R^{+}...
-
Amortization of Intangibles For each of the following intangible assets , indicate the amount of amortization expense that should be recorded for the year 2010 and the amount of accumulated...
-
McGown Corporation has the following information: Raw Materials Inventory Work in Process Inventory Finished Goods Inventory Additional information for the year is as follows: Raw materials purchases...
-
An investor accumulated 1800 shares of Microtel Corporation over a period of several months. She bought 1000 shares at $15.63, 500 shares at $19.00, and 300 shares at $21.75. What was her average...
-
When engaging in monetary policy, the impact of expansionary policy on an aggregate demand aggregate supply model is to a. Increase aggregate demand. b. Increase aggregate supply. c. Decrease...
-
The elasticity of demand is related to the slope of the demand curve A. And only the slope of the demand curve. B. But also the (price, quantity) position on the demand curve. C. But also the slope...
-
The adjusted trial balance of the Jee, Moe, and Ole partnership at December 31, 2011, is as follows: Cash ................................................. $50,000 Accounts receivable-net...
-
The option adjusted spread (OAS) of a callable bond is zero. Is it true that this callable's yield equals that of the noncallable counterpart? Explain your answer.
-
The 0.5-year and 1-year risk free spot interest rates are 5% and 6% per year. Assume semi-annual compounding. Compute the 0.5- and 1-year zero coupon bond prices that pays $1 face value at maturity.
-
Expected return on Metal's stock is 16%. Market risk premium and the expected return on the market portfolio are 8% and 14%, respectively. Metal's stock has a beta of 1.25. Is Metal's stock...
-
There are two retailers, A, B. A had a price of $717.93 and (forward) EPS of $9.78. B had a price of $31.21 and (forward) EPS of $3.77. Calculate (forward) P/E ratios
-
Assume that r f = 10%; E (r m ) = 16%and the return on stock C is 18%. Determine the implicit beta for stock C.
-
Middleton Co. paid $80,000 cash to purchase land from Saws Lumber Company. Saws originally paid $80,000 for the land. Required a. Did this event cause the balance in Middletons cash account to...
-
1. Use these cost, revenue, and probability estimates along with the decision tree to identify the best decision strategy for Trendy's Pies. 2. Suppose that Trendy is concerned about her probability...
-
Explain which compound has the higher melting point or boiling point: a) Melting point b) Boiling point or C COCH c) Boiling point or
-
What is the functional group present in these compounds? SCH3 SH C CH, - Ph b) ) H3 f) CH,CH,SOH d) CH,CH,SPh e) CH,SCH3
-
Identify the most acidic site in thesecompounds: NH2 CH.COCH,CH,CH3 c) a) b) CH3 e) CH;CH,CH,COH d) CH,CH,CCH,CH3
-
1. A businessman travels between the cities of the following graph. The travel pattern is that he stays in one of the cities for the whole day, and on the next day, he travels to one of the...
-
How do historical legacies of colonialism, imperialism, and systemic injustice continue to shape contemporary power structures, and what steps can be taken to address and redress these historical...
-
Izabella is 20 years old. Eighteen months ago, she started selling some of her old clothing which no longer suited her on peer-to-peer social shopping apps such as Letgo and Depop. She was surprised...

Study smarter with the SolutionInn App


