Starting with acetylene as your only source of carbon atoms, propose a plausible synthesis for 1, 4-dioxane:
Question:
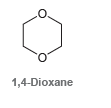
Transcribed Image Text:
1,4-Dioxane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
H Lindlars ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using acetylene as your only source of carbon atoms, design a synthesis of trans-5- decene:
-
Using acetylene as your only source of carbon atoms, design a synthesis of cis-3- decene:
-
Using acetylene as your only source of carbon atoms, design a synthesis of pen-tanal. (Pentanal has an odd number of carbon atoms, while acetylene has an even number of carbon atoms): H.
-
East Corp is a mature company and expects its sales and operating costs to be fixed (or unchanged) in perpetuity. It has sales of $524,000 and operating costs of $272,480. The firm has a tax rate of...
-
a. In how many ways can two sorted arrays of N elements be merged? b. Give a nontrivial lower bound on the number of comparisons required to merge two sorted lists of N elements, by taking the...
-
Provide detailed proofs of the trace identities: (a) traces of products of \(\gamma\) matrices (with no \(\gamma^{\mu \dagger}\) ) are representation independent (same for \(\gamma^{\mu}\) and...
-
This exercise considers the data described in the Section 13.2.2 ratemaking classification example using data in Table 13.3 Table 13.3 . a. Fit a gamma regression model using a log-link function with...
-
Mississippi Manufacturing, Inc., reported the following at December 31, 2014 and December 31, 2015: Stockholders Equity Preferred stock, cumulative, $2.00 par, 6%, 70,000 shares issued .... $ 140,000...
-
Think about TWO clients with different needs and describe their situation. For each one, select a range of current resources available for providing information, advice or guidance within a specific...
-
Consider the composite solid shown. Solid A is a thermally conductive material that is 0.5-cm thick and has a thermal conductivity, KA = 50 W=m K. The back side of solid A (x = 0) is thermally...
-
Assume a DVC and an IAC presently have real per capita outputs of $500 and $5000, respectively. If both nations have a 3 percent increase in their real per capita outputs, by how much will the per...
-
True or False: Some economists argue that the single best thing that IACs could do for DVCs in terms of economic growth would be to eliminate trade barriers between IACs and DVCs.
-
TopChop sells hairstyling franchises. TopChop receives $50,000 from a new franchisee for providing initial training, equipment and furnishings that have a stand-alone selling price of $50,000....
-
Marc attended University of Wisconsin for two years and earned his Bachelor of Science degree. Assume that Mother lives 40 years and dies with an estate of $100,000. Her only heirs are her three...
-
We're looking at the dawn of modern mathematics. Focus on connections between the 17th century and Indian mathematics when discussing the 17th century. Modern mathematics is modeled on Greek...
-
A contractor wants to buy a piece of equipment to use over 5 years and then sell it. The equipment cost $25,000. It provides an annual revenue of $8,000 and incurs annual expenses of $2,000. At the...
-
DE Assume the on-campus demand for soda is as shown in the following table. Calculate total revenue and marginal revenue and then answer two questions about price in a perfectly competitive market...
-
Of all the book fines a library issued one year, 0.8% of them were for amounts greater than $10. There were 15 fines greater than $10. How many fines did the library issue that year? Enter your...
-
Lucille wants to install an irrigation system to water all six of her flowerbeds in her backyard. The drawing shows the location of each flowerbed and the distance between them in feet. (a) Represent...
-
The polar coordinates of a point are given. Find the rectangular coordinates of the point. (-1, - /3)
-
Imidazole forms part of the structure of the amino acid histidine and can acts as both an acid and a base (a) Look at the electrostatic potential map of Imidazole, and identify the most acidic...
-
Of the two vitamins A and C, and is hydrophilic and water-soluble while the other is hydrophobic and fat-soluble. Which iswhich? CH CH- CH CH2OH H3C CH3 H- CH Vitamin A (retinol) Vitamin C...
-
Fill in the multiple bonds in the following model of naphthalene, C10H8 (gray = C, ivory =H). How many resonance structures does naphthalenehave?
-
1. Mention five key organizational characteristics that the Cll research team identified for organizations with effective quality management systems. provide brief discussion of each and how it...
-
When a metal was exposed to photons at a frequency of 1.46 1015 s1, electrons were emitted with a maximum kinetic energy 3.60 10-19 J. G ? Calculate the work function, , of this metal. J/photon What...
-
No. 101 Debit Cash $ 2,400 Question 2 (10 pts) On November 1, 2024, the account balances of Schilling Equipment Repair were as follows. Accumulated Depreciation-Equipment A Clipboard Font Alignment...

Study smarter with the SolutionInn App


