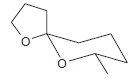
The compound below is believed to be a wasp pheromone. Draw the major product formed when this
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
H3...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The compound below is an example of a methyl ester. Methyl esters react with lithium iodide to give lithium carboxylate salts. The solvent in this example is pyridine (margin). Suggest several...
-
Identify all of the products formed when the compound below is treated with aqueous acid: N- excess H,O+
-
A material is believed to be a compound. Suppose you have several samples of this material obtained from various places around the world. Comment on what you would expect to find upon observing the...
-
Determine whether each relation defines y as a function of x. Give the domain. Identify any linear functions. y = |x|
-
Use the triangle at the right. Find the length of the missing side. Show your work. 1. a = 16, b = 63 2. b = 2.1, c = 2.9 3. A 10-foot ladder leans against a wall with its foot braced 3 feet from...
-
A company can borrow funds at an after-tax cost of 4.5%. The companys stock price is \($40\) per share, earnings per share is \($2.00\), and the company has 15 million shares outstanding. If the...
-
Consider Example 3.5. For each of the 10 boys, we randomly assigned the left or right sole to material A and the remaining side to B. Use $R$ 's sample function to simulate a treatment assignment....
-
Viola Holloway and Renee Forrow are the sole equal shareholders in the corporation of Holloway And Forrow Enterprises, Inc. The Ilolloway And Eomow Enterprises, Inc. owns the following depreciable...
-
TB MC Qu. 3-53 (Algo) Listed below are year-end account balances... Listed below are year-end account balances ($ in millions) taken from the records of Symphony Stores. Debit Credit Accounts...
-
240 cm 3 of ethane (C 2 H 6 ) was burnt in a controlled way and found to raise the temperature of 100 cm 3 of water by 33.5 C. (specific heat capacity of water = 4.18Jg 1 K 1 ; 1mol of gas molecules...
-
Propose an efficient synthesis for each of the following transformations: (a) (b)
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OMe Meo Meo Br Br
-
The half-life of radioactive potassium is 1.3 billion years. If 10 grams is present now, how much will be present in 100 years? In 1000 years?
-
Study Appendix 13. Consider the following data for the Rivera Company: From the preceding information, fill in the following blanks. Be sure to mark your variances. F for favorable and U for...
-
The cost structure of a manufacturer of micro- chips is described in the table that follows. The firm's fixed costs equal $10,000 per day. Calculate the average variable cost, average fixed cost, and...
-
When do you use soft approaches to problem solving in your day-to-day life? Why do you use them?
-
Name and describe the per se violations of antitrust law.
-
In Problem 26-14, what is the value of the Herfindahl- Hirschman Index? Problem 26-14 Suppose that a company based in Dallas, Texas, confronts only four other rival firms. Its own market share is 35...
-
Describe the role of inflation in cosmology. How does it help to explain why the geometry of the universe is flat? What is the source of the energy that drove the rapid expansion of the universe...
-
On the basis of the details of the following fixed asset account, indicate the items to be reported on the statement of cashflows: ACCOUNT Land ACCOUNT NO. Balance Date Item Debit Credit Debit Credit...
-
Give the free - redical chain mechanism for the formation of ethy l bromide from ethane and bromine in the presence of light.
-
Answer Problem 8.33 for all alcohols with the forrnula C4H9OH. (a) Give the structures of all alcohols with the molecular formula C5H13OH. (b) Which of the compounds in part (a) are chiral? (c) Name...
-
Withc-rut consulting tables, arrange the compounds within each of the following sets in order of increasing boiling point, and give your reasoning. (a) I -hexanol, 2-pentanol, tert-butr,l alcohol (b)...
-
Suppose you invest 52%, 28%, and 20% of your wealth into a stock, the market, and a risk-free asset, respectively. The beta of the stock is 1.1. What is the beta of the portfolio?
-
An investment of $21745, earning compound interest, grows by $2278 in one year. At this rate of growth, how long will it take for the original investment to double?
-
On September 12, 3,400 shares of Denver Company's common stock are acquired at a price of $58 per share plus a $170 brokerage commission. On October 15, an $1.20-per-share dividend was received on...

Study smarter with the SolutionInn App


