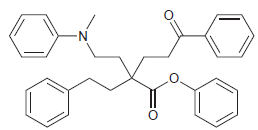
The following compound has four aromatic rings. Rank them in terms of increasing reactivity toward electrophilic aromatic
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
A B N Increasing ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following compound has two isomers: ClCH==CHCl One isomer has a dipole moment of 0 D, and the other has a dipole moment of 2.95 D. Propose structures for the two isomers that are consistent with...
-
The following compound has only one asymmetric carbon. Why then does it have four stereoisomers? CH CH CHCH,CH CHCH Br
-
The following compound has been found to be an inhibitor of penicillinase. The enzyme can be reactivated by hydroxylamine (NH2OH). Propose a mechanism to account for the inhibition and for the...
-
For problems involving composite bodies composed of two or more materials, the elasticity solution requires both boundary conditions and interface conditions between each material system. The...
-
Use a search engine to locate the most recent "E-mail Marketing Trends Survey." Using the information in it, work with a team of your classmates to select a local small business with which you are...
-
Is each of the following a method used in linkage, cytogenetic, or physical mapping? A. Fluorescence in situ hybridization (FISH) B. Conducting two-factor crosses to compute map distances C....
-
What is a double-barreled question?
-
Central Ski and Cycle purchased 50 pairs of ski boots for $360 per pair less 33 % and 10%. The regular rate of markup on selling price of the boots is 40%. The stores overhead is 22% of the selling...
-
Newly formed S&J Iron Corporation has 196,000 shares of $6 par common stock authorized. On March 1, Year 1, S&J Iron issued 10,000 shares of the stock for $11 per share. On May 2, the company issued...
-
There are 2 shinobis with chakra levels 5 and 10 respectively and the desired sum of chakra levels is utmost 15 Starting with ke0, suy of chakra levels after attack max(5-0,0) + max(10- 0,0) 5+10 15....
-
The following compound has two aromatic rings. Identify which ring is expected to be more reactive toward an electrophilic aromatic substitution reaction.
-
For each compound below, identify which position(s) is/are most likely to undergo an electrophilic aromatic substitution reaction. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH3 NO2 O,N. CH3 O,N NO2
-
Let F(x, y, z) = (3x 2 yz - 3y) i + (x 3 z - 3x) j + (x3y + 2z) k. Evaluate C F dr, where C is the curve with initial point (0, 0, 2) and terminal point (0, 3, 0) shown in the figure. z. ZA (0, 0,...
-
New York license plates consist of three letters followed by three numerals, and 245 letter arrangements are not allowed. How many plates can New York issue?
-
Self-employed persons can make contributions for their retirement into a special tax-deferred account called a Keogh account. Suppose you are able to contribute \(\$ 20,000\) into this account at the...
-
Find the sum of the odd integers between 48 and 136 .
-
Suppose that you expect to receive a \(\$ 100,000\) inheritance when you reach 21 in seven years and four months. What is the present value of your inheritance if the current interest rate is \(1.4...
-
Evaluate the expressions in Problems 11-18. \(\sum_{k=3}^{5} k\)
-
Write the equation of a sine function that has the given characteristics. Amplitude: 3 Period: /2 Phase shift: 2
-
The overall reaction and equilibrium constant value for a hydrogenoxygen fuel cell at 298 K is 2H 2 (g) + O 2 (g) 2H 2 O(l) K = 1.28 10 83 a. Calculate E cell and G 8 at 298 K for the fuel cell...
-
Five isomeric alkenes A-E each undergo catalytic hydrogenation to give 2-methylpentane. The IR spectra of these five alkenes have the following key absorptions (in cm-1): Compound A: 912 (s), 994...
-
From the information in Table 12.3, predict the appearance of the molecular ion peak(s) in the mass spectrum of chloromethane. (Assume that the molecular ion is the base peak.) Table 12.3 TABLE 12.3...
-
Indicate whether the following peaks in the mass spectrum of 1-heptanol are odd-electron or even-electron ions. (a) m/z = 83 (b) m/z = 70 (c) m/z = 56 (d) m/z = 41
-
Can you elaborate on the processes of somatic hypermutation and class-switch recombination in B lymphocytes, detailing their roles in generating antibody diversity and optimizing antibody-mediated...
-
Can you delve into the complex interplay between the gut microbiota and host immune system, highlighting the mechanisms by which commensal microbes shape immune development, tolerance, and...
-
Marisa wants to buy a home in Atlanta with a 30-year mortgage that has an annual interest rate of 4.9%. The house she wants is $250,000, and she will make a $55,000 down payment and borrow the...

Study smarter with the SolutionInn App


