Question: The following compound is optically inactive. Explain why. CH3 H3C-
The following compound is optically inactive. Explain why.
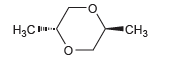
"CH3 H3C-
Step by Step Solution
3.31 Rating (157 Votes )
There are 3 Steps involved in it
This compound has a center of inver... View full answer

Get step-by-step solutions from verified subject matter experts


