The triphenylmethyl radical reacts with itself to form the following dimer: Identify the type of radical process
Question:
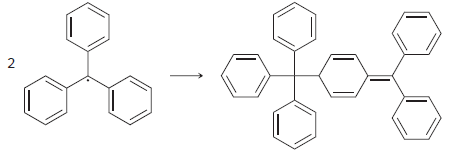
Identify the type of radical process taking place, and draw the appropriate fishhook arrows.
Transcribed Image Text:
2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The triphenylmethyl radical was the first radical to be observed. Draw all resonance structures of this radical, and explain why this radical is unusually stable:
-
Cyclopentadiene (C5H6) reacts with itself to form dicyclopentadiene (C10H12). A 0.0400 M solution of C5H6 was monitored as a function of time as the reaction 2 C5H6 C10H12 proceeded. The following...
-
The triphenylmethyl cation is so stable that some of its salts can be stored for months. Explain why this cation is so stable. triphenylmethyl cation
-
How to respond to the following? Cycle stock, pipeline inventory, buffer stock, and in-transit inventory all have associated carrying costs. Interest rates on inventory values, warehousing expenses,...
-
Paul's Medical Equipment Company manufactures hospital beds. Its most popular model, Deluxe, sells for $5,000. It has variable costs totaling $2,800 and fixed costs of $1,000 per unit, based on an...
-
The country with the highest sustainable sales growth is: A . Norvolt. B . Abuelio. C . Borliand.
-
Describe a variety of ways in which medical records have been falsified.
-
From the information in BE5-3, prepare the journal entries to record these transactions on Rodriguez Companys books under a perpetual inventory system.
-
1. Corporate Finance deals with raising funds for Private Companies, Corporate entities and Governments. What are these funds not used for ? Explain
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
What is the House of Quality matrix and how is it used?
-
Show that (C V /V) T = T( 2 P/ T 2 ) V
-
In what situations would you expect to model a relationship between an agent and a resource?
-
4. (1pt) You just purchased a zero coupon bond from the Treasury for $950.00. The bond has a face value of $1,000 and matures in 5 years. What is the yield you will earn if you hold the bond until...
-
Which of the following is not a requirement for spousal support payments to be deductible? a. The amount for child support must be specified separately. O b. The payments must be made for a period of...
-
8 0 , 0 0 0 lb / h of a 1 0 % soda solution in water will be concentrated to 5 0 % . Water vapor available at 1 0 0 psia and 3 4 0 F will be used. The third evaporator has a pressure of 2 psia. The...
-
Sky Train Inc. originally issued 10-year bonds with a face value of $1000 at par. The bonds have a coupon rate of 8%, and coupons are paid semiannually. The bonds will mature in 6 years, and the...
-
Simplify 3x (2x-3x+4) (3x-4)(2x+3) 3x(2x) (2x-1)(3x-3x+4)
-
What kind of nuclear power is responsible for sunshine?
-
CRUZ, INC. Comparative Balance Sheets December 31, 2015 CRUZ, INC. Income Statement For Year Ended December 31, 2015 Required Use the indirect method to prepare the cash provided or used from...
-
Draw -D-galactopyranose and -D-mannopyranose in their more stable chair conformations. Label each ring substituent as either axial or equatorial. Which would you expect to be more stable, galactose...
-
Draw -L-galactopyranose in its more stable chair conformation, and label the sub-stituents as either axial or equatorial.
-
Identify the following monosaccharide, write its hill name, and draw its open-chain form in Fischerprojection.
-
Write a short report on a (recent) development that affects the audit market, the audit profession and audit quality. In this report, you are asked to 1) Briefly describe the development and its link...
-
How do cutting-edge technologies such as artificial intelligence and blockchain facilitate organizational flexibility, enabling dynamic resource allocation, process optimization, and rapid...
-
How can individuals cultivate cognitive flexibility and emotional resilience amidst the complexities of modern life, navigating intricate personal and professional dynamics to thrive in rapidly...

Study smarter with the SolutionInn App


