Using 1-propanol as your only source of carbon, propose an efficient synthesis for each of the following
Question:
(a)
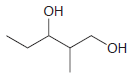
(b)
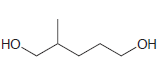
(c)
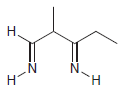
Transcribed Image Text:
ОН ОН но но.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
a b c OH ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using 2-propanol as your only source of carbon, show how you would prepare 2-methyl-2-pentanol.
-
Using acetylene as your only source of carbon atoms, design a synthesis of pen-tanal. (Pentanal has an odd number of carbon atoms, while acetylene has an even number of carbon atoms): H.
-
Using acetylene as your only source of carbon atoms, identify a synthetic route for the production of 1-bromobutane.
-
What is the present value of $9,000 received: a. Twenty eight years from today when the interest rate is 10% per year? b. Fourteen years from today when the interest rate is 10% per year? c. Seven...
-
The risk-free rate, KRF, is 6 percent and the market risk premium, (KM - KRF), is 5 percent. Assume that required returns are based on the CAPM. Your $1 million portfolio consists of $700,000...
-
Your linker uses a single pass through the set of given object files to find and resolve external references. Each object file is processed in the order given, all external references are found, and...
-
Describe a variety of negligent errors that have application to all healthcare professionals.
-
Hatcher Carriage Company offers guided horse- drawn carriage rides through historic Columbus, Georgia. The carriage business is highly regulated by the city. Hatcher Carriage Company has the...
-
Bodin Company manufactures finger splints for kids who get tendonitis from playing video games. The firm had the following inventories at the beginning and end of the month of January. January 1...
-
Auto Supply Company's 2018 statement of cash flows appears in Exhibit 13-8. Study the statement and respond to the following questions. In Exhibit 13-8 a. What was the company's free cash flow in...
-
Refer to the illustrative example of Chapter 7 where we fitted the Cobb Douglas production function to the manufacturing sector of all 50 states and the District of Columbia for 2005. The results of...
-
Refer to Example 7.4. For this problem the correlation matrix is as follows: a. Since the zero-order correlations are very high, there must be serious multicollinearity. Comment. b. Would you drop...
-
Philadelphia Fastener Corporation manufactures nails, screws, bolts, and other fasteners. Management is considering a proposal to acquire new material-handling equipment. The new equipment has the...
-
What is the purpose of the notes to the financial statements?
-
BRK Limited is being sued for $3.5 million by a client for defamation resulting from statements made in newspapers by one of its executives. In each of the situations below, state whether a liability...
-
For each situation below, state whether an asset would be recognised in the balance sheet. If the answer is no, state which of the essential characteristics relating to assets has not been met. 1....
-
The information is based on the spreadsheet as shown Suppose Ingredient I is made up of \(80 \%\) micoden and \(20 \%\) water, Ingredient II is made up of \(30 \%\) micoden, \(50 \%\) bixon, and \(20...
-
The sum of the birth years of actresses Debra Winger and Meryl Streep is 3,904. If Debra is six years younger, in what year was she born?
-
Suspend an open-topped container of water in a pan of boiling water, with its top above the surface of the boiling water. Youll note that although water in the inner container can reach 100C, it cant...
-
Find the volume of the described solid S. A frustum of a right circular cone with height h, lower base radius R, and top radius r -r- --R
-
Complete the reactions given in Fig. P21.52 by giving the principal organic products. Explain how you arrived at your answers. NaOH CH O (trace) H,C CCHO CH +CH OH (solvent) Ph NH2 1 (CgH,NO3)...
-
Identify each of the following compounds from their spectra. (a) Compound A: molecular mass 113; gives a positive hydroxamate test; IR 2237, 1733, 1200 cm-1; proton NMR: 1.33 (3H, t, J = 7 Hz), ...
-
Outline a synthesis of each of the following compounds from the indicated starting materials and any other reagents. (a) 1-cyclohexyl-2-methyl-2-prupanol from bromocyclohexane (b) PhNHCH2CH2CH(CH3)2...
-
A business provides services valued at 1 1 0 0 and sends an invoice to the client. The client has promised to pay in one month. which two accounts will be affected?
-
Rushing's financial statements were finalized on March 3, 2025. Required: 1. What amount(s) if any, related to the situations described should Rushing report among current liabilities in its balance...
-
Monroe Company produces and sells three products. Information regarding these three products for 2024, the most recent year, is provided below: selling price per unit Product #1 $26 variable costs...

Study smarter with the SolutionInn App


