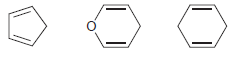
Which of the following compounds would you expect to be most acidic? Justify your choice.
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Deprotonatio...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds would you expect to show ultraviolet absorptions in the 200 to 400nmrange? (a) (b) (c) CN CH3 (f) (d) (e) " N. Indole Aspirin
-
Which of the following compounds would you expect to have a ? ? ?* UV absorption in the 200 to 400 nm range? (a) (b) (c) (CH3)2C=C=Do CH2 A ketene Pyridine
-
Which of the following compounds would you expect to exhibit only London forces? a. Potassium chloride, KCl b. c. Silicon tetrafluoride, SiF4 d. Phosphorus pentachloride, PCl5 (
-
Use the trapezium rule, with interval-halving and extrapolation, to evaluate 0 log(cosh x) dx to 4dp
-
What is markup? How is it used to determine prices?
-
Describe the rationale underlying the technique of an electrophoretic mobility shift assay.
-
Describe how you can use www.yellowpages.com to create a sample in the state of New York by using systematic sampling technique.
-
Jack Herron is an IT auditor with McGee LLP, a large national public accounting firm. His manager, Amanda McDermott, has assigned him to the Linz Company audit. The McGee financial auditors have...
-
Reddy Company is a merchandising firm. Next month the company expects to sell 500 units. The following data describe the company's revenue and cost structure: Selling price per unit. $60 Sales...
-
Martin Shipping Lines issued bonds ten years ago at $1,000 per bond. The bonds had a 30-year life when issued, with semiannual ayments at the then annual rate of 10 percent. This return was in line...
-
What are some things Amazon does to manage inventories in their warehouses?
-
How can performance measures sometimes cause the wrong things to happen?
-
Construct an example of a 4 4 unitary matrix.
-
Use a calculator to evaluate an ordinary annuity formula \[A=m\left[\frac{\left(1+\frac{r}{n}ight)^{n t}-1}{\frac{r}{n}}ight]\] for \(m, r\), and \(t\) (respectively) given in Problems 7-22. Assume...
-
The following extracts relating to suppliers and accounts payable are taken from the annual report of the John Lewis Partnership. Customers, products and suppliers (Extract from Business Review, p....
-
A \(\$ 1,000,000\) lottery prize pays \(\$ 50,000\) per year for the next 20 years. If the current rate of return is \(2.25 \%\), what is the present value of this prize?
-
A bag contains 12 pieces of candy. In how many ways can five pieces be selected?
-
Use a calculator to evaluate the present value of an annuity formula \[P=m\left[\frac{1-\left(1+\frac{r}{n}ight)^{-n t}}{\frac{r}{n}}ight]\] for the values of the variables \(m, r\), and \(t\)...
-
Find the exact value of expression. Do not use a calculator. cos 540 - tan(-405)
-
The following information is available for Partin Company: Sales $598,000 Sales Returns and Allowances 20,000 Cost of Goods Sold 398,000 Selling Expense 69,000 Administrative Expense 25,000 Interest...
-
(a) From what Grignard reagent can 3-methl-l pentanol be prepared by reaction with ethylene oxide, then aqueous acid? (b) Give the structure of another epoxide and another higher-order curpate that...
-
Explain why all attempts to isolate trimethyloxonium iodide lead instead to methl iodide and dimethl ether.
-
Explain why all attempts to isolate trimethyloxonium iodide lead instead to methl iodide and dimethl ether.
-
Based on the introductory in Public Finance, what do you think must be the main role of the government in our individual lives? Any references will do, as long as the answer is related to question...
-
Apercu Industries borrowed $92,350 for 200 days at 15% simple interest. Find the total interest they will pay (a) using bankers' rule and (b) using the exact method.
-
On January 4th, Mark went to the hospital to have an appendectomy. His deductible is $2,500 and since it is the beginning of the year, he has not contributed anything to the deductible yet. The total...

Study smarter with the SolutionInn App


