Write an equation for the proton transfer reaction that occurs when each of the following acids reacts
Question:
(a) HBr
(b)
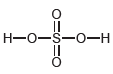
(c)
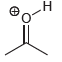
(d)
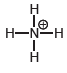
Transcribed Image Text:
Н-о—$—0—н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a b c...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A test developer wishes to collect evidence about the construct validity of a specific assessment instrument. To accomplish this, a multitrait-multimethod technique is applied. This resuits in the...
-
Write an equation for the reaction between ethylene oxide and a. CH3CH2CH2CH2MgCl followed by hydrolysis b. C6H5CH2MgBr followed by hydrolysis c. H2C=CHLi followed by hydrolysis d. CH3CC-Na+ followed...
-
Write an equation for the reaction between a. Ethylmagnesium iodide and water b. Phenylmagnesium bromide and ethanol
-
Clark had brain surgery. Insurance will not pay for the surgery until the deductible of $1,000 is hit. Then Clark's coinsurance of 80% / 20% kicks in. Clark has an out-of-pocket maximum of $7,500....
-
Respond to the following: 1. Leading and Lagging Indicators a. Answer the question: Which is more useful, explaining what happened yesterday or predicting what is likely to happen tomorrow? Explain....
-
Congressman Snowball, having read Chapter 11 of this text, informs his colleagues that people who have higher levels of education have higher earnings capacities. He argues, based on Chapter 17, that...
-
Some of the mathematical steps in Example 5.2 are omitted. Derive Eqs. (5.14), (5.17), and (5.18). Data From Example 5.2:- Figure 5.10:- Data From Eqs. 5.14 , 5.17, and 5.18:- Herbicides such as...
-
Companies need to offer competitive benefits to employees or risk having employees become dissatisfied with their current job situations. KPMG Canada recently faced such a challenge when the companys...
-
"How can an organization leverage automated backup verification processes to ensure the reliability and recoverability of backup data? Discuss the potential role of machine learning and artificial...
-
a) Given matrices A, U, E and V, write a pseudocode to determine if UEV is the SVD of A. You may use the function [E, F] = eigs(X) to determine the eigenvectors E corresponding to the eigenvalues in...
-
Methylene chloride (CH 2 Cl 2 ) has fewer chlorine atoms than chloroform (CHCl 3 ). Nevertheless, methylene chloride has a larger molecular dipole moment than chloroform. Explain.
-
Suggest an efficient synthesis for each of the following transformations: a. b.
-
(a) For each data set, find the mean, median, and mode. (b) Discuss anything about the data that affects the usefulness of each statistic as a measure of center. a. Class absences (12 students): 0,...
-
With the growth of free trade, Mexican employers have sought to reduce union control over internal labor markets, and they have eliminated promotion by seniority, rules against subcontracting, and...
-
It has been observed that unions in the capital-intensive steel industry were able to negotiate higher-than-average wage increases during the very period in which steel output in the United States...
-
A certain country has very centralized collective bargaining, under which wage bargains are applied nationally. This country is thinking about adopting a bargaining structure that is more...
-
The following table gives the demand for labor at two different firms. The current wage rate in both firms is $7 per hour. A union would like to organize employees in one of the firms and bargain to...
-
Suppose that the demand for rough laborers is L D = 100 - 10W, where W = the wage in dollars per hour and L = the number of workers. If immigration increases the number of rough laborers hired from...
-
Determine the following indefinite integrals. dx x + 1
-
Splitting hairs, if you shine a beam of colored light to a friend above in a high tower, will the color of light your friend receives be the same color you send? Explain.
-
Show the products of thesereactions: CH Br NaBH4 LIAIH4 ether b) a) CH,OH CH3
-
Show the products of thesereactions: CH,CI 1) NANH2, NH3 (/), NACN b) CH,C3C- a) 2) CH,CH,CH,Br DMSO Br NaCN c) DMSO 1) NANH, 2) CH,I I) NANH2, NH, (1) d) HC=CH 2) CH,CH,Br DMF Br + I NACN e) Cl- Br...
-
Suggest methods for preparing these compounds from alkylhalides: CN CH3 c) CH;C=CCH Ph b) HC=CCH,CH,CHCH3 a) "CH3
-
In January 2023, Marty's Fine Pens, a business carried on as a sole proprietorship, sells a limited-edition fountain pen for $125,000. The cost of the pen is $63,000. There is a down payment of...
-
An 90000 loan is amortized by payments of $1850 at the end of every 6 months at a rate of 2% compounded monthly 1. Construct a partial amortization schedule showing the last 2 payments 2. determine...
-
Kelso's has a return on equity of 15.2 percent, a debt-equity ratio of 44 percent, a capital intensity ratio of 1.08, a current ratio of 1.25, and current assets of $138,000. What is the profit...

Study smarter with the SolutionInn App


