(a) Two (unnormalized) excited state wavefunctions of the H atom are Normalize both functions to 1. (b)...
Question:
(a) Two (unnormalized) excited state wavefunctions of the H atom are
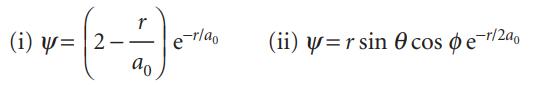
Normalize both functions to 1.
(b) Confirm that these two functions are mutually orthogonal.
Transcribed Image Text:
(i) u = 2 ਹੈ % -r/ao (ii) y=r sin cos e-r/2a0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What are the units of the H atom total energy eigenfunctions? Why is a 0 3/2 R(r) graphed in Figure 20.6 rather than R(r)?
-
A hydrogen atom is excited from its ground state to the state with n = 4. (a) How much energy must be absorbed by the atom? Consider the photon energies that can be emitted by the atom as it...
-
Two unnormalized functions of the hydrogen atoms are (2)2 1 1 2 2 sincos 2 Where N1 and N2 are the normalization constants The volume element for spherical polar coordinate is 2 sin and the limits...
-
Neutrons can be used in diffraction experiments to probe the lattice structure of crystalline solids. Since the neutron's wavelength needs to be on the order of the spacing between atoms in the...
-
You are given two paired samples with the following information: Based on these paired samples, test at the = 0.05 level whether the true median paired difference is 0. 2 le 3 4 O 2 1 5 9 438...
-
Convection heat transfer coefficients for a heated horizontal surface facing upward may be determined by a gage whose specific features depend on whether the temperature of the surroundings is known....
-
The monthly maintenance charges of an organization are normally distributed, with a mean of \($500\) and a standard deviation of \($150.\) Find the probability that a randomly selected utility bill...
-
Kingston, Inc. management is considering purchasing a new machine at a cost of $4,133,250. They expect this equipment to produce cash flows of $814,322, $863,275, $937,250, $1,017,112, $1,212,960,...
-
Cullumber Inc. manufactures cycling equipment. Recently, the vice president of operations of the company has requested construction of a new plant to meet the increasing demand for the company's...
-
Gonzalez Electric Company has outstanding a 10 percent bond issue with a face value of $1,000 per bond and three years to maturity. Interest is payable annually. The bonds are privately held by...
-
Determine which of the following functions are eigenfunctions of the inversion operator (which has the effect of making the replacement x x): (a) X 3 kx, (b) Cos kx, (c) X 2 + 3x 1. State the...
-
Calculate the average linear momentum of a particle described by the following wavefunctions: (a) E ikx , (b) Cos kx, (c) E x2 , where in each one x ranges from to +.
-
Non-uniform displacement-current density figure 32-30 shows a circular region of radius R = 3.00 cm in which a displacement current is directed out of the page. The magnitude of the density of this...
-
B.2. Consider the given relational schema Intern(internid, name) Job(internid, depid) Department(deptid, supervisor) Write the relational algebra, Tuple relational Calculus and SQL for the given...
-
CONSOLIDATED STATEMENTS OF EARNINGS, COMPREHENSIVE INCOME AND RETAINED EARNINGS - USD ($) $ in Millions Income Statement [Abstract] Sales Cost of sales (without depreciation) Depreciation Gross...
-
What are the potential benefits of employee empowerment as a means of decreasing staff turnover and increasing customer satisfaction?
-
(a) Explain the concept of Ordered Binary Decision Diagrams (OBDDS) and list examples of their applications in high-level synthesis. [5 marks] (b) Represent the Boolean function f(a,b,c) = abc + abc...
-
Question: - Based on the article links given above, create a complete detailed summary that clearly indicates the reasons why the USD appreciated or depreciated relative to the foreign currency....
-
a. What is capital budgeting? b. What is the difference between independent and mutually exclusive projects? c. 1. Define the term net present value (NPV). What is each franchise's NPV? 2. What is...
-
Avatar Financials, Inc., located on Madison Avenue, New York City, is a company that provides financial advice to individuals and small- to mid-sized businesses. Its primary operations are in wealth...
-
Verify that the radius ratio for (a) sixfold coordination is 0.414, and (b) for eightfold coordination is 0.732.
-
In an X-ray investigation, the following structure factors were determined (with F h00 =F h00 ): Construct the electron density along the corresponding direction. h F1,00 0 10 1 -10 2 8 3 -8 4 6 5 -6...
-
What are the values of the angle of the first three diffraction lines of bcc iron (atomic radius 126pm) when the X-ray wavelength is 72pm?
-
You are the vice president for support services at Sunrise Nursing and Rehabilitation Center. Frances "Frankie" Hammerman, the full-time coordinator of volunteers, reports to you. She has held that...
-
How do the tenets of contemporary neuropsychoanalysis reconcile the historically divergent perspectives of Freudian psychoanalytic theory and modern neuroscientific understandings of brain function ?
-
1. Define the term "core competencies" and their significance in shaping a company's competitive advantage 2. Provide TWO (2) examples of companies that have effectively leveraged their core...

Study smarter with the SolutionInn App


