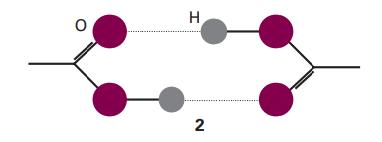
Acetic acid vapour contains a proportion of planar, hydrogen bonded dimers (2). The apparent dipole moment of
Question:
Acetic acid vapour contains a proportion of planar, hydrogen bonded dimers (2). The apparent dipole moment of molecules in pure gaseous acetic acid has a magnitude that increases with increasing temperature. Suggest an interpretation of this observation.
Transcribed Image Text:
H 2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
Explanation The dipole moment of a molecule is a measure of the electric charge distribution ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Acetic acid vapour contains a proportion of planar, hydrogen-bonded dimers. The relative permittivity of pure liquid acetic acid is 7.14 at 290K and increases with increasing temperature. Suggest an...
-
Gaseous acetic acid molecules have a certain tendency to form dimers. (A dimer is a molecule formed by the association of two identical, simpler molecules.) The equilibrium constant Kc at 25oC for...
-
Gaseous acetic acid molecules have a certain tendency to form dimers. (A dimer is a molecule formed by the association of two identical, simpler molecules.) The equilibrium constant Kp at 25oC for...
-
1 30 2 3 4 If A= = -1 2 1,B= 1 23 0 02 -1 1 2 5 9 13 (a) -1 2 4 -12 4 1 24 (C) -1 2 4 -2 24 then AB= 5 (b) -1 9 13 24 -2 2 4 (d) None of these
-
The flowchart for Problem 1 illustrates two alternative inventory ordering methods. Required a. Distinguish between a purchase requisition and a purchase order. b. Discuss the primary advantage of...
-
Some nickel(ii) sulfate solution was carefully placed in the bottom of a beaker of water. The beaker was then covered and left for several days. a. Describe what you would see after: (i) A few hours...
-
Reconsider Parts (a) through (f) of Problem 2. For each "true" statement, develop a mathematical proof based on the time value of money factor equations from Table 2.6 in Chapter 2. Data from problem...
-
Inventoriable CostsError Adjustments Werth Company asks you to review its December 31, 2010, inventory values and prepare the necessary adjustments to the books. The following information is given to...
-
An 80 kg person stands in an elevator which is ascending towards the top of a building. A) As the elevator begins its ascent, it accelerates at the rate of 1 m/s2 . Draw the free body diagram showing...
-
Give three examples from the MIPS architecture of each of the architecture design principles: (1) simplicity favors regularity (2) make the common case fast (3) smaller is faster (4) good design...
-
Draw examples of the arrangements of electrical charges that correspond to monopoles, dipoles, quadrupoles, and octupoles. Suggest a reason for the different distance dependencies of their electric...
-
Plot the magnitude of the electric dipole moment of hydrogen peroxide as the HeOeOeH (azimuthal) angle changes from 0 to 2. Use the dimensions shown in 1. H 97 pm O 149 pm 1
-
The formulas of three noncyclic hydrocarbons are listed below. Which of these is an alkene? (a) C 4 H 10 (b) C 3 H 4 (c) C 5 H 10
-
How do disruptive technologies such as artificial intelligence, quantum computing, and biotechnology drive transformative innovation ecosystems, reshaping industries, and redefining the boundaries of...
-
Compare and contrast the characteristics of monopolistically competitive, monopolistic, and perfectly competitive markets. Provide an organization that is an example of each and discuss how changes...
-
Cadbury Celebrations All over the world, chocolate is one product that gets almost uniform recognition. This is the product which is normally targeted at the younger age groups across the world....
-
International organizations respond to global social injustice by providing a variety of services that save lives and build futures. Some organizations provide relief aid such as food, shelter, and...
-
What role do protected areas, wildlife reserves, and marine sanctuaries play in safeguarding biodiversity hotspots, critical habitats, and key biodiversity areas, and how can networks of protected...
-
Let f : [a, b] R be C1 on [a, b] with f(t) 0 for t [a, b]. Prove that the explicit curve x = f-1(y), as y runs from f(a) to f(b), is orientation equivalent to the explicit curve y = f(x), as x...
-
The slopes of the tangents at the points where the curve y = x2 - 4x intersects the X-axis is 1) 1 2) +2 3) +3 4) +4
-
Assume that a system has a very large number of energy levels given by the formula l = 0 l 2 with 0 = 1.75 10 -22 J, where l takes on the integral values 1, 2, 3, . . Assume further that the...
-
Determine in each of the following cases if the function in the first column is an eigenfunction of the operator in the second column. If so, what is the eigenvalue? a. x 2 ..x 2 / 8 d 2 /dx 2 b. x 3...
-
Is the function 2x 2 1 an eigenfunction of the operator (3/2 x 2 ) (d 2 /dx 2 ) + 2x (d /dx)? If so, what is the eigenvalue?
-
A student holds a ruler between their thumb and index finger, suddenly releases it , and then as fast as possible, captures it again, measuring the distance it has dropped. they repeat this procedure...
-
A 0.140-kg baseball is dropped and reaches a speed of 1.20 m/s just before it hits the ground and bounces. It rebounds with an upward velocity of 1.00 m/s. What is the change of the ball's momentum...
-
A small office building air conditioner operates on 250 V AC and consumes 3.78 kW. (a) What is its effective resistance? (b) What is the cost of running the air conditioner during a hot summer month...
Sustainable Development An Appraisal From The Gulf Region 1st Edition - ISBN: 1785333534 - Free Book

Study smarter with the SolutionInn App


