Question: Consider the arrangement shown in 5 for a system consisting of an O-H group and an O atom, and then use the electrostatic model of
Consider the arrangement shown in 5 for a system consisting of an O-H group and an O atom, and then use the electrostatic model of the hydrogen bond to calculate the dependence of the molar potential energy of interaction on the angle θ.
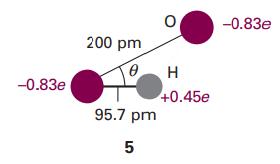
-0.83e 200 pm 0 95.7 pm 5 H +0.45e -0.83e
Step by Step Solution
3.57 Rating (157 Votes )
There are 3 Steps involved in it
The potential energy of interaction between two molecules is given by Uq1q24... View full answer

Get step-by-step solutions from verified subject matter experts


