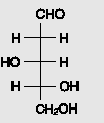
Examine the structure of deoxyribose, part of the backbone in DNA. Does deoxyribose exhibit optical isomerism? If
Question:
Transcribed Image Text:
Сно н -н но -н Н Он Cн-ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
With two chiral ce...View the full answer

Answered By

Sufiyan Ahmed Tariq
I am a Chartered Accountant and an Associate Public & Finance Accountant. I also hold a bachelors of Commerce degree. I have over 8 years of experience in accounting, finance and auditing. Through out my career, I have worked with many leading multinational organisation.
I have helped a number of students in studies by teaching them key concepts of subjects like accounting, finance, corporate law and auditing. I help students understanding the complex situation by providing them daily life examples.
I can help you in the following subject / areas:
a) Accounting;
b) Finance;
c) Commerce;
d) Auditing; and
e) Corporate Law.
4.90+
7+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the structure of deoxyribose and number the carbon atoms. Describe the numbering of the carbon atoms in deoxyribose with regard to the directionality of a DNA strand. In a DNA double helix, what...
-
Examine the structure of cyclosporin A (page 503). a. By drawing a dashed line at each peptide bond, deduce how many amino acid units are present in cyclosporin A. b. Three of the units are...
-
Indicate whether each of the following statements about the structure of DNA is true or false. (Each letter is used to refer to the concentration of that base in DNA.) (a) A + T = G + C (b) A = G; C...
-
Review the following exhibits for Kitchenmaid: (Click the icon to view the functional cost breakdown.) (Click the icon to view the customer feature ratings.) m link the innn in this site function...
-
Tots Ltd manufactures babies' play suits for sale to retail stores. All play suits are of the same design. There are two departments: the cutting department and the machining department. You are...
-
Gary Weiss is uncertain about the steps used to prepare a production cost report. State the procedures that are required in the sequence in which they are performed.
-
A company issues a \(10 \%\) coupon bond that matures in 5 years. However, this company is in trouble, and it is estimated that each year there is a probability of .1 that it will default that year....
-
Financial ratio analysis is conducted by managers, equity investors, long-term creditors, and short-term creditors. What is the primary emphasis of each of these groups in evaluating ratios?
-
2.) What is the specific goal of this lab? Exactly what question are you trying to answer? Be as specific as possible. ("To learn about topic X..." is not specific!) 3.) What specific measurements or...
-
Two firms are competing on a market with demand P-47-3Q by simultaneously choosing what price to charge. Assume they can charge any real price (not just limited to integer numbers) Whoever charges...
-
Name the alkene: CHs- CH-CH-- CH2-CHa
-
Show the tripeptide that result from linking the amino acids glycine, valine, and serine in that order. Label the N-terminus and C-terminus ends of the tripeptide. | N - -- HN --- -- HN-C-- ...
-
Define percentile rank.
-
A 3.0-cm-diameter sphere is maintained at 1000C and has an emissivity of 0.6. It is enclosed by another sphere having a diameter of 9.0 cm with an emissivity of 0.3 (inside and outside). Both spheres...
-
Question 1: For a uniform thin square plate of mass m, determine its mass moment of inertia about the y axis, which passes through its gravitational center G as shown. L y G y G Profile view (a) (b)...
-
Gadget Ltd. is considering to go under liquidation procedures after the global effects of corona virus on the industry. It just announced today will be paying an annual dividend totally $7 million...
-
For the project depicted in the decision tree below: a. What is the expected profit for the project and should the project be pursued if the MARR is 8%? b. What should be the strategy? Show all...
-
What are the reaction forces Rz , Ry , and Mx for both supports c) N a = 250 mm M Support IV 200 000 Nmm b = 1000 mm Support V c = 250 mm F = 1000 N
-
Write the key accounting equation on which balance sheets are based. What are the most important bank assets? What are the most important bank liabilities?
-
Explain the Hawthorne effect.
-
Classify each change as physical or chemical. a. Natural gas burns in a stove. b. The liquid propane in a gas grill evaporates because the valve was left open. c. The liquid propane in a gas grill...
-
Classify each property as physical or chemical. a. The boiling point of ethyl alcohol b. The temperature at which dry ice evaporates c. The tendency of iron to rust d. The color of gold
-
Classify each property as physical or chemical. a. The tendency of ethyl alcohol to burn b. The shine on silver c. The odor of paint thinner d. The flammability of propane gas
-
GDP exceeds NDP by an amount equal to? Explain
-
A not-for-profit hospital reported in 2021 income statement: Revenue 150m Expenses 110m Net Income 40m On their 2021 Statement of Changes in Net Assets they reported: Beginning Net Assets of 110m...
-
Various cost accounting methods record, classify, analyze, and evaluate alternative courses of action used to control costs. The chief information security officer (CISO) uses these results to...

Study smarter with the SolutionInn App


