Explain the direction of the chemical shifts for Fe(0), Fe(II), and Fe(III) in Figure 22.20. Fe 2p
Question:
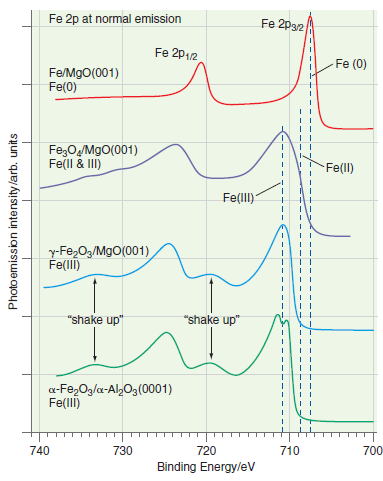
Transcribed Image Text:
Fe 2p at normal emission Fe 2p32 Fe 2p12 Fe (0) Fe/MgO(001) Fe(0) Fe;OM9O(001) Fe(ll & II) Fe(ll) Fe(lI) y-Fe,O3/MgO(001) Fe(llI) "shake up" "shake up" a-Fe,Og/a-Al2Og (0001) Fe(llI) 740 730 720 710 700 Binding Energy/ev Photo emission intensity /arb. units
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
As electrons are removed from Fe to form Fe 2 or Fe 3 ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the chemical shifts of the protons in the following compounds. (a) (b) (c) (d) (e) (f) HC(CH3)3 CH CH CH3 , - CH,CCCH OH CH,CH,-C-OH CH3 CHC-CH Br
-
The proton NMR spectrum of 2-pyridone gives the chemical shifts shown. (a) Is 2-pyridone aromatic? (b) Use resonance forms to explain your answer to (a). Also explain why the protons at (7.31and...
-
The chemical shifts of the 1H nuclei in 2,2-dimethylpropane and TMS are 0.95 and 0.0, respectively. From these data, what can you deduce about the relative electronegativities of carbon and silicon?
-
A non reactive/conservative contaminant is dumped on the ground level and it leaches to the groundwater vertically and takes half day for reaching the groundwater by travelling through unsaturated...
-
The point is on the terminal side of an angle in standard position. Find the exact values of the six trigonometric functions of the angle. 1. (12, 16) 2. (7, -24) 3. (-0.5, 4.5)
-
a. What fact orsled to Toyotas leadership position in auto mobile quality? b. What was the purpose of CCC21? c. What evidence does the case present of Toyotas managerial arrogance? d. What should...
-
You are purchasing a new car. The possible manufacturers, car sizes, and colors are listed in the table. How many different ways can you select one manufacturer, one car size, and one color? Use a...
-
Meridian Clothing is a retail store specializing in womens clothing. The store has established a liberal return policy for the holiday season in order to encourage gift purchases. Any item purchased...
-
Set out below are the draft statements of profit or loss of Smiths and its subsidiary company Flowers for the year ended 31 December 20X7. On 1 January 20X6 Smiths purchased 75,000 ordinary shares in...
-
The following is part of a TCP header dump (contents) in hexadecimal format. E293 0017 00000001 00000000 5002 07FF a. What is the source port number? b. What is the destination port number? c. What...
-
The principal line in the emission spectrum of sodium is yellow. On close examination, the line is seen to be a doublet with wavelengths of 589.0 and 589.6 nm. Explain the source of this doublet.
-
Why is XPS a surface-sensitive technique?
-
Take a look at solved problem SI. Let's suppose a male is heterozygous for two polymorphic sequence-tagged sites. STS-1 exists in two sizes: 211 bp and 289 bp. STS-2 also exists in two sizes: 115 bp...
-
Identify some of the kinds of terms individuals may want to include in a premarital agreement that may not be enforceable and explain why. In this context, explain the importance of a severability...
-
What is plain carbon steel? Give the classification of plain carbon steels and their important properties and uses.
-
Identify at least ten ethical dos that should guide the professional conduct of paralegals.
-
Define elasticity, rigidity, hardness, fatigue, ductility, brittleness.
-
Identify the most commonly included provisions of a premarital agreement.
-
Assuming that f is differentiable for all x, simplify f(x) f(25) lim 5
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
The heat capacity of chloroform (trichloromethane, CHCl 3 ) in the range 240 K to 330 K is given by C p,m /(J K 1 mol 1 ) = 91.47 + 7.5 10 2 (T/K). In a particular experiment, 1.00 mol CHCl 3 is...
-
A sample consisting of 1.0mol CaCO 3 (s) was heated to 800C, when it decomposed. The heating was carried out in a container fitted with a piston that was initially resting on the solid. Calculate the...
-
A chemical reaction takes place in a container of cross-sectional area 100 cm 2 . As a result of the reaction, a piston is pushed out through 10 cm against an external pressure of 1.0 atm. Calculate...
-
What is a plan on how you intend to be successful in future semesters. The plan should be a source of motivation to do well in college, both professionally and personally. Include tips that you can...
-
Question 1. From the research for this terms Crisis Management Team Assignment - Taylor swift and Ticket Master Crisis a. Explaining, with some detail, three (3) specific things that the leader(s) in...
-
If individual values that underline ethics are developed at a young age, what might this suggest about the potential for ethical conflicts to arise within an organization?

Study smarter with the SolutionInn App


