Explain why the fluorescence and absorption groups of peaks in Figure 25.10 are shifted and show mirror
Question:
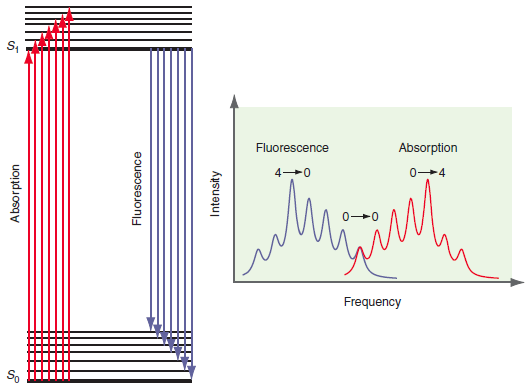
Transcribed Image Text:
Fluorescence Absorption 0-4 Frequency So Absorption Fluorescence Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
The absorption event is initiated from the n 0 vibrational level of the ground state The rela...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The fluorescence spectrum of anthracene vapour shows a series of peaks of increasing intensity with individual maxima at 440 nm, 410 nm, 390 nm, and 370 nm followed by a sharp cut-off at shorter...
-
1. G. Johnson and colleagues have analyzed the MAP kinase cascade in which MEKK2 participates in mammalian cells. By a yeast two-hybrid screen (see Chapter 7), MEKK2 was found to bind MEK5, which can...
-
What is the difference between a fluorescence excitation spectrum and a fluorescence emission spectrum? Which one resembles an absorption spectrum?
-
The conjugate base of diethyl malonate can serve as a nucleophile to attack a wide range of electrophiles. Identify the product that is formed when the conjugate base of diethyl malonate reacts with...
-
A guy wire runs from the ground to a cell tower. The wire is attached to the cell tower 150 feet above the ground. The angle formed between the wire and the ground is 43° (see figure). (a) How...
-
Clips Online sells subscriptions for \($36\) for 12 months. The company collects cash in advance and then subscribers have access to unlimited movies each month. Apply the revenue recognition...
-
Describe the forms of evidence presented by a plaintiff at trial.
-
Following is a traditional income statement for Mouse Max, a company that manufactures cordless mice for computers. Revenue ................................... $2,500,000 Cost of goods sold...
-
J. Clancy owns Clancy Consulting. On December 31 of the prior year, the Retained Earnings account balance was $27,000. During the current year, the company paid $13,800 in cash dividends. Clancy...
-
Canyon Canoe Company has experienced rapid growth in its first few months of operations and has had a significant increase in customers renting canoes and purchasing T-shirts. Many of these customers...
-
What aspect of the confocal microscope makes single-molecule spectroscopy in solutions possible?
-
The rate of fluorescence is higher than that for phosphorescence. Can you explain this fact?
-
What are the benefits to be gained by knowing that a proposed sample is more than 5% of the total populations size? In what marketing situation might this be a common occurrence?
-
Explain the terms supply chain and logistics.
-
Jean Kelley owned three limited partnership interests in 2018: How much is her passive loss deduction (against nonpassive activities) and suspended loss for each activity? 2018 Allocated Date Gain/...
-
Describe some of the obstacles to lean success.
-
Evaluate the studies which aim to establish the link between SHRM and organisational performance.
-
Analyse the relationship between organisational structure and SHRM.
-
In problem, find the domain of the composite function f g. 2 8(x) f(x) = x + 3'
-
Which of the following raises the credibility of areport? Which of the following raises the credibility of a report? Multiple Choice avoiding predictions avoiding the use of cause-effect statements...
-
Calculate the maximum (zero-current) potential difference of a nickelcadmium cell, and the maximum possible power output when 100 mA is drawn at 25C.
-
Derive an expression for the current density at an electrode where the rate process is diffusion-controlled and c is known. Sketch the form of j/j L as a function of c . What changes occur if anion...
-
Can magnesium be deposited on a zinc electrode from a unit activity acid solution at 25C?
-
Assume that a firm has weighted average cost of capital of 3.6% with a corporate tax rate of 21%. The pre-tax cost of debt is 3.1%. In case the firm would have no debt, the cost of equity for the...
-
Consider the stock of Aya Company, which will pay an annual dividend of $3 one year from today. The dividend will grow at a constant rate of 3%, forever.The market requires a 12% return on the...
-
Analyze the video presented on the link below. Also, please discuss or explain the video and relate it to personal finance. https://www.youtube.com/watch?v=nFY0HBkUm8o

Study smarter with the SolutionInn App


