Question: Figure 16.17 shows that atomic level resolution is only attainable in the repulsive portion of the tipsurface potential. What does this tell you about the
Figure 16.17
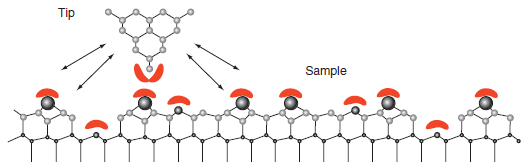
Tip Sample
Step by Step Solution
3.40 Rating (163 Votes )
There are 3 Steps involved in it
The observation shows that the repulsive part ... View full answer

Get step-by-step solutions from verified subject matter experts


