For many enzymes, the mechanism of action involves the formation of two intermediates: Show that the rate
Question:
For many enzymes, the mechanism of action involves the formation of two intermediates:

Show that the rate of formation of product has the same form as that shown in eqn 20H.1, but with vmax and KM given by
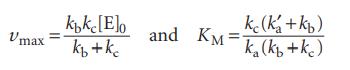
Data in eqn 20H.1
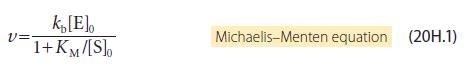
Transcribed Image Text:
E+S →ES ES →E+S ES ES' ES' → E+P v=k₂[E][S] v=k₁ [ES] v=k₁[ES] v=k [ES']
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
solution The rate of formation of product in the given enzymatic rea...View the full answer

Answered By

Brian Nambale
As someone who loves to read books and discuss ideas with friends, I have developed a strong foundation of good knowledge that allows me to understand complex topics well, making me confident in my ability to deliver subjects effectively when teaching children.Engaging in thought-provoking discussions with friends helps me deepen my understanding and apply good thinking to expand my knowledge in various subjects, which I am eager to well when teaching children.During my college years, I acquired 5 years of tutoring experience, which not only helped me to support myself financially but also provided me with an opportunity to share my knowledge, inspire students, and make a positive impact on their lives. Through this experience, I developed strong teaching and communication skills, which have served me well throughout my academic and professional journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Describe the mechanism of action for addictive drugs such as cocaine.
-
The mechanism of glycoside formation is the same as the second part of the mechanism for acetal formation. Propose a mechanism for the formation of methyl -D-glucopyranoside.
-
What's the mechanism of action of trizepatide, the new diabetes 2 drug? and how does it differ from GLP-1 agonists?
-
Why should every credit application be signed by the applicant or an authorized officer of the applicant? There are several factors that influence the setting of the terms and conditions of a sale....
-
A written hospital policy prohibited solicitation and distribution in all patient care areas, including hallways adjacent to patients rooms and other areas frequented by patients. The policy...
-
a. $20 000 is invested in an account that pays interest at 2.4% per annum. The interest is added at the end of each year. After how many years will the value of the account first be greater than $25...
-
Plaintiff sought to enforce against the defendant estate a promise made by his now-deceased uncle to pay Plaintiff a sum of money if Plaintiff refrained from the use of alcohol and tobacco for a...
-
Grace Herron has just approached a venture capitalist for financing for her new business venture, the development of a local ski hill. On July 1, 2013, Grace was loaned $150,000 at an annual interest...
-
If the trend towards authoritarianism continues and more democracies move towards the totalitarian side of the spectrum, how could that affect the international political system?
-
Luna Company is an IFRS reporter. Luna Company acquired 100% of the voting stock of the AutoMania Group on January 1 of the current year for a total acquisition cost of $251,000. The trial balance of...
-
The mechanism of a composite reaction consists of a fast preequilibrium step with forward and reverse activation energies of 27 kJmol 1 and 35 kJmol 1 , respectively, followed by an elementary step...
-
The rate of a chemical reaction is found to triple when the temperature is raised from 24 C to 49 C. Determine the activation energy.
-
Northern Energy Limited ( NEL) is a large Canadian private company organized in three operating segments: propane operations, trucking, and mineral explorations. Financial statements have not been...
-
In the case of a change in accounting principle, the entity must Multiple select question. compute the cumulative effect of change on the preceding 10 years tax liability. disclose the number of...
-
You are evaluating buying an apartment building. It is expected to generate annual cash flow of $100,000 for each of the next 6 years. Please use this information for problems 3 - 5. If you have to...
-
Which of these amended the Truth in Lending Act, requiring credit and charge card issuers to provide certain disclosures in direct mail, telephone, and other applications and solicitations for...
-
An ordinary annuity assumption means that cash is expected to change hands Multiple Choice in a series of exchanges made at the beginning of each period. in a series of exchanges made at the end of...
-
Debtor, whose income is below the median in her state, is driving a two-year-old Lexus RX 350, which is a small luxury sport utility vehicle. Debtor does not drive the vehicle off the road, but feels...
-
a) Let a < b and c < d be real numbers. Sketch a graph of the rectangle [a, b] [c, d] := [(x, y) : x [a, b], y [c, d]}, and decide whether this set is compact or connected. Explain your answers....
-
Government is advised to tax goods whose demand curves are inelastic if the goal is to raise tax revenues. If the goal is to discourage consumption, then it ought to tax goods whose demand curves are...
-
Calculate the expectation value (r r) 2 if the H atom wave function is 100(r).
-
In spherical coordinates, z = r cos. Calculate z and z 2 for the H atom in its ground state. Without doing the calculation, what would you expect for x and y, and x 2 and y 2 ? Why?
-
The force acting between the electron and the proton in the H atom is given by F = e 2 / 4 0 r 2 . Calculate the expectation value F for the 1s and 2p z states of the H atom in terms of e, 0 , and a...
-
Image transcription text PART ONE You are provided with the following data for Kasama Ltd and Choma Ltd. KASAMA LTD Boom Normal Recession Year 2020 2021 2020 2021 2020 2021 End of Year Price (K) 20...
-
Alonzo works as a car detailer. Charge $175 per car. He is planning to move out of his parents' house and rent his first apartment. He will be required to pay $120 for application fees, $950 for...
-
Pola Sommers, a massage therapist, needs 5 ounces of a 22% lavender oil solution. She has only 4% and 40% lavender oil solutions available. How many ounces of each should Pola mix to obtain the...

Study smarter with the SolutionInn App


