For some proteins, the isoelectric point must be obtained by extrapolation because the macromolecule might not be
Question:
For some proteins, the isoelectric point must be obtained by extrapolation because the macromolecule might not be stable over a very wide pH range. Estimate the pH of the isoelectric point from the following data for a protein:
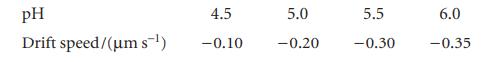
Transcribed Image Text:
pH Drift speed/(um s-¹) 4.5 -0.10 5.0 -0.20 5.5 -0.30 6.0 -0.35
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (13 reviews)
The isoelectric point is the pH at which a molecule has a net charge of zero This ...View the full answer

Answered By

Dansteve Matoke
As a consequence of more than three years of experience in ACADEMIC WRITING, I have vast, diverse knowledge and impeccable grammar. I guarantee the quality of my work across multiple fields. I intend to precisely meet the clients' expectations. Let's work together to achieve exceptional grades.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For some proteins, the isoelectric point must be obtained by extrapolation because the macromolecule might not be stable over a very wide pH range. Estimate the pH of the isoelectric point from the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
1. The Jarmon Company manufactures and sells a line of exclusive sportswear. Prepare a financial position as at 31st December 2022 from the following information. Account Receivable Long term loan...
-
Nationwide Mutual Insurance, based in Columbus, Ohio, is one of the largest diversified insurance and financial services organizations in the world, with more than $140 billion in assets. Nationwide...
-
Why are preference decisions sometimes called rationing decisions?
-
Consider PowerBar, the strategy for which is summarized in the PowerBar case. What implications for the culture, structure, systems, and people would you suggest given the nature of the product and...
-
Accelerator, Inc., manufactures a fuel additive, Surge, that has a stable selling price of $44 per drum. The company has been producing and selling 80,000 drums per month. In connection with your...
-
When a company lends money to employees at a rate of 4%, the company will record ______. Multiple choice question. a liability called Accounts Payable a liability called Notes Payable an asset called...
-
Redstone Mill is a manufacturer that makes all sales on 30-day credit terms. Annual sales are approximately $40 million. At the end of 2014, accounts receivable were presented in the company's...
-
At 20C the diffusion coefficient of a macromolecule is found to be 8.3 10 11 m 2 s 1 . Its sedimentation constant is 3.2 Sv in a solution of density 1.06 g cm 3 . The specific volume of the...
-
Discuss the physical origins of the surface Gibbs energy.
-
Evaluate the following integrals and show details of your work.
-
Indicate the effect (increase, decrease, no change, or not enough information) that each of the following situations has on break-even unit sales. If you answer 'not enough information', list the...
-
Describe the operating cycle of a retail business. How are the operating cycles of a retail business and a service business similar to and different from each other?
-
When developing a selling expenses budget and a general and administrative expenses budget, why do you have to know how expenses behave?
-
Why is having too much cash on hand a problem?
-
If the total variable cost per unit increases while the selling price per unit, the fixed costs and the sales volume remain the same, how would you expect the change in variable costs to affect...
-
What is payroll outsourcing? When might a company consider outsourcing its payroll?
-
Draw the major product for each of the following reactions: (a) (b) (c) 1) 9-BBN 2) H2O2, NaOH 1) Disiamylborane 2) H20, NaOH
-
Determine whether the integral over /J and /, in Exercise 12.15a is zero over a symmetrical range about e = 0 in the group C3v
-
List the symmetry elements of the following molecules and name the point groups to which they belong: (a) Staggered CH3CH3, (b) Chair and boat cyclohexane, (c) B, H6, (d) [Co (en) 3J3+, where en is...
-
The group D 1h has a C, axis perpendicular to the principal axis and a horizontal mirror plane. Show that the group must therefore have a centre of inversion.
-
A 33-year-old male patient returns from a 6-year tour of navy duty where he was stationed in Thailand. The patient presents with impairment of proprioception and vibratory sensation along with loss...
-
Several complaints were received from residents on over speeding in a newly constructed road. A team of transportation engineers carried out a site visit. After conducting a survey (under controlled...
-
Explain why felons should vote First, felons should be allowed to vote because it is their right as American citizens. It is their responsibility as citizens who have paid their debts to society and...

Study smarter with the SolutionInn App


