Identify the molecular orbitals for F 2 in the images shown here in terms of the two
Question:
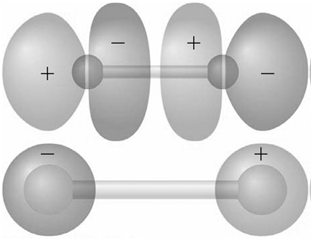
Transcribed Image Text:
+ +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
The top image has symmetry the AOs are out of ...View the full answer

Answered By

Elias Gichuru
am devoted to my work and dedicated in helping my clients accomplish their goals and objectives,providing the best for all tasks assigned to me as a freelancer,providing high quality work that yields high scores.promise to serve them earnestly and help them achieve their goals.i have the needed expertise,knowledge and experience to handle their tasks.
4.80+
325+ Reviews
859+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Draw the molecular orbitals for the cyclopropenyl case. (Since there are three p orbitals, there must be three MOs: one all-bonding MO and one degenerate pair of MOs.) (b) Draw an energy diagram...
-
Evaluate the Klopp case in terms of those two criteria.
-
Images of molecular orbitals for LiH calculated using the minimal basis set are shown here. In these images, the smaller atom is H. The H1s AO has a lower energy than the Li2s AO. The energy of the...
-
One of Red Clay's employee handbook sections covers employee monitoring. An employee filed a complaint with the HR department stating he opposes this monitoring and chooses to opt out. What legal...
-
Different types of granular substances naturally settle at different angles when stored in cone-shaped piles. This angle θ is called the angle of repose (see figure). When rock salt is...
-
Walking along the beach, you notice that a new wave reaches the shore every \(4.0 \mathrm{~s}\), and you estimate the wave crests to be \(2.5 \mathrm{~m}\) apart. At what wave speed \(c\) are the...
-
Describe what is meant by retaliatory and constructive discharge and how to defend a claim for unfair discharge.
-
Xavier Stores Company and Lestrade Stores Inc. are large retail department stores. Both companies offer credit to their customers through their own credit card operations. Information from the...
-
The shaft of a gearbox is coupled through a No. 80 chain drive, rotating a driveshaft for a pulp screen at a paper-producing plant. The screen driveshaft rotates at 200 rpm clockwise. The drive...
-
You have designed and created tables for the City Jail database. These tables dont include any constraints. Review the information to determine what constraints you might need for the City Jail...
-
Explain why the nodal structures of the 1 g MOs in H 2 and F 2 differ.
-
In discussing Figure 23.2, the following statement is made: Interchanging red and blue does not generate a different MO. Justify this statement. Figure 23.2 H1s His H2 Energy
-
Muskoka Canoes has had a defined benefit pension plan for three decades. Two years ago, the company improved the benefits at a cost of $2,800,000. Pension plan assets were $84,000,000 while pension...
-
Describe the disallowed deductions and losses of corporations.
-
Mara transferred assets to Con Co. on May 15, 2018. Mara acquired the assets on June 15,2015 . What is Con Co.'s holding period for the assets?
-
How do corporations treat start-up expenditures?
-
Jason is single and uses the calendar year for his tax year. He died on May 15, 2018. When is his final income tax return due?
-
To qualify for the immediate expensing election, an asset must meet what qualifications?
-
Between 12:00 PM and 1:00 PM , cars arrive at Citibanks drive-thru at the rate of 6 cars per hour (0.1 car per minute). The following formula from statistics can be used to determine the probability...
-
Willingness to pay as a measure of a person's value for a particular good measures the maximum a person would be willing to pay requires that payment actually be made depends on the satisfaction that...
-
Discuss the origins of diagonal and cross peaks in the COSY spectrum of an AX system.
-
Calculate the frequency separation of the nuclear spin levels of a 13 C nucleus in a magnetic field of 14.4 T given that the magnetogyric ratio is 6.73 10 7 T 1 s 1 .
-
33 S has a nuclear spin of 3/2 and a nuclear g-factor of 0.4289. Calculate the energies of the nuclear spin states in a magnetic field of 7.500 T.
-
Hall & Company issues $ 1 0 0 , 0 0 0 , 0 0 0 in 6 % bonds with detachable warrants of 1 0 warrants for each $ 1 , 0 0 0 bond. The bonds are issued at 9 9 . Record the issuance under the following...
-
Assume that hein acquired Dolan Development last year. Hein recorded the following intangible assets on the date of acquisition: Goodwill: $ 1 , 5 0 0 , 0 0 0 Dolan Development trademark: $ 6 0 0 , 0...
-
What is the total amount of interest that will be paid of the loan is carried to term with no extra payments made building cost 3 3 0 0 0 0 . loan started im Dec 2 0 2 3 worh a 7 . 5 % interest rate...

Study smarter with the SolutionInn App


