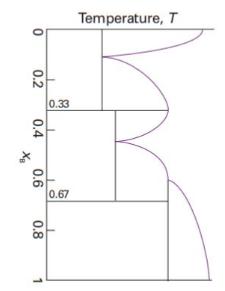
Label the regions of the phase diagram in Fig. 6.38. State what substances (if compounds give their
Question:
Label the regions of the phase diagram in Fig. 6.38. State what substances (if compounds give their formulas) exist in each region. Label each substance in each region as solid, liquid, or gas.
Fig. 6.38.
Transcribed Image Text:
Temperature, T 0 T 0.2 0.33 0.4 X₂ 0.6 0.67 0.8 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
Answer Region A Solid ...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Label the regions of the phase diagram in Fig. 6.3 7. State what substances (if compounds give their formulas) exist in each region. Label each substance in each region as solid, liquid, or gas.
-
The phase diagram of a hypothetical substance is (a) Estimate the normal boiling point and freezing point of the substance. (b) What is the physical state of the substance under the following...
-
In most compounds the solid phase is denser than the liquid phase. Why isnt this true for water?
-
4. This problem investigates nearest neighbor and bilinear interpolation. For simplicity, we will focus on estimating the image intensity at a single location. Interpolation is used when transforming...
-
Explain the difference between an exchange and an OTC market.
-
Based on the dimensionless conduction heat rates for cases 12-15 in Table 4.1 b, find shape factors for the following objects having temperature T1, located at the surface of a semi-infinite medium...
-
Waste gas \(\left(\mathrm{CO}_{2} ight)\) is vented to outer space from a spacecraft through a circular pipe \(0.2 \mathrm{~m}\) long. The pressure and temperature in the spacecraft are \(35...
-
The Giles Agency offers a 12% trade discount when providing advertising services of $1,000 or more to its customers. Audreys Antiques decides to purchase advertising services of $3,500 (not including...
-
1. Identify a specific business that uses Artificial Intelligence and Robotics. 2. Why did you select this organization for your research project? 3. Identify three major issues for the use of...
-
A U.S. company owns an 80% interest in a company located on Mars. Martian currency is called the Martian Credit. During the year the parent company sold inventory that had cost $24,000 to the...
-
What factors determine the number of theoretical plates required to achieve a desired degree of separation in fractional distillation?
-
Haemoglobin, the red blood protein responsible for oxygen transport, binds about 1.34 cm 3 of oxygen per gram. Normal blood has a haemoglobin concentration of 150 g dm 3 . Haemoglobin in the lungs is...
-
Evaluate [(-3) + 7 ] - (-4).
-
In several well-written paragraphs, describe what it means for an Agile Team to be Self-Organizing and Cross-Functional. Please address the characteristics of being "Self-Organizing" and...
-
7. The expected return on the Market Portfolio M is E(RM)=15%, the standard deviation is OM-25% and the risk-free rate is R=5%. a. Suppose that stock X has standard deviation x=30%, and correlation...
-
Marketing success depends on developing a differentiated offering that addresses a need for one or more target markets, then positioning that offering effectively in the minds of target customers....
-
How does Eggers (Jeffrey Eggers TED talk ) describe our expectations of leaders and leadership? Do you agree? Why? How could Complex Adaptive Leadership possibly address the conditions Eggers...
-
Question 3. Compressed air stored at ambient temperature is used as part of a cooling system. A nozzle, with an exit area of 22 mm2 delivers the air. Air is to be delivered into a change with ambient...
-
Is the set of vectors {(1, 1, 1), (0, 1, 1,), (0, 0, 1)} R3 linearly dependent?
-
Briefly discuss the implications of the financial statement presentation project for the reporting of stockholders equity.
-
List the symmetry elements of the following molecules and name the point groups to which they belong: (i) NO 2 , (ii) N 2 O, (iii) CHCl 3 , (iv) CH 2 =CH 2 .
-
State the symmetry criteria that allow a molecule to be optically active.
-
What is the maximum degeneracy of a particle confined to the interior of an octahedral hole in a crystal?
-
Gator Incorporated reported taxable income of $1,500,000 this year and paid federal income taxes of $500,000. Included in the company's computation of taxable income is a $98,500 gain from the sale...
-
Comparative data from the statement of financial position of Ridgewood Ltd. are shown below. 2024 2023 2022 Current assets $1,537,000 $1,182,000 $1,217,000 Property, plant, and equipment 3,101,000...
-
Alexis has trouble closing sales with new customers but not with existing customers. What advice would you give Alexis?

Study smarter with the SolutionInn App


