Some polymers can form liquid crystal mesophases with unusual physical properties. For example, liquid crystalline Kevlar (3)
Question:
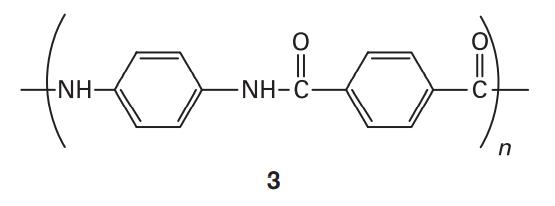
Some polymers can form liquid crystal mesophases with unusual physical properties. For example, liquid crystalline Kevlar (3) is strong enough to be the material of choice for bulletproof vests and is stable at temperatures up to 600 K. What molecular interactions contribute to the formation, thermal stability, and mechanical strength of liquid crystal mesophases in Kevlar?
Transcribed Image Text:
O formam -NH- -NH-C 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Solution Liquid crystal Kevlar is a network polymer with molecula...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Some polymers can form liquid crystal mesophases with unusual physical properties. For example, liquid crystalline Kevlar (1) is strong enough to be the material of choice for bulletproof vests and...
-
Some polymers have unusual properties. For example, Kevlar (3) is strong enough to be the material of choice for bulletproof vests and is stable at temperatures up to 600K. What molecular...
-
The American Aluminum Company is considering making a major investment of $150 million ($5 million for land, $45 million for buildings, and $100 million for manufacturing equipment and facilities) to...
-
Joe rents his condo for $1,500 per month. Total rental and personal use days for the current year was 210 days and 20 days, respectively. What are the tax consequences for Joe?
-
What is the OTC market for trading derivatives? How do OTC markets differ from exchanges?
-
An isothermal silicon chip of width W = 20 mm on a side is soldered to an aluminum heat sink (k = 180 W/m K) of equivalent width. The heat sink has a base thickness of Lb = 3 mm and an array of...
-
Uniform horizontal sheets of water of 3-mm thickness issue from the slits on the rotating manifold shown in Fig. P12.3. The velocity relative to the arm is a constant \(3 \mathrm{~m} / \mathrm{s}\)...
-
Life cycle product costing. Intentical Inc., manufactures game systems. Intentical has decided to create and market a new system with wireless controls and excellent video graphics. Intenticals...
-
McGyver runs a hot air balloon company in Liverpool that takes customers on flights. He hears that Murdock has inherited a hot air balloon from his aunt Penny. McGyver asks to inspect the balloon,...
-
Building One Properties is a limited partnership formed with the express purpose of investing in commercial real estate. The firm is currently considering the acquisition of an office building that...
-
Hexane and perfluorohexane show partial miscibility below 22.70C. The critical concentration at the upper critical temperature is x = 0.355, where x is the mole fraction of C 6 F 14 . At 22.0C the...
-
Refer to the information in Exercise 6.15(b) and sketch the cooling curves for liquid mixtures in which x(B 2 H 6 ) is (a) 0.10, (b) 0.30, (c) 0.50, (d) 0.80, and (e) 0.95. Data in Exercise 6.15(b)...
-
Record the following transactions of Fashion Designs in a general journal: DATE TRANSACTIONS 2013 April 2 Sold merchandise for cash, $1,000 plus sales tax. 3 The customer purchasing merchandise for...
-
Nadya Trojanowski, CEO of the new start-up Ayuda.ai, which develops an AI assistant for household tasks, seeks to raise $1.5 million in a private placement of equity in her early-stage venture. Nadya...
-
Can J.C. Penney Be Saved? Apply the knowledge of management presented in this chapter to the following case. The goal of this case analysis is to enable you to understand what happened at J.C. Penney...
-
What is cost of quality? Describe the total quality cost. Company A spends $ 5 0 , 0 0 0 on quality on inspection and receive $ 6 0 , 0 0 0 as complaint. Company B spends $ 1 0 , 0 0 0 on quality...
-
a. Explain the Reynold Transport Theorem and the difference between a control volume and system. b. In the image below, a pump can spray water at two powers: A and B. Sketch control volumes for each...
-
"Financial intermediaries have evolved to reduce transaction costs and allow small savers and borrowers to benefit from the existence of financial markets." Examine this statement. (7 marks) Analyse...
-
What are the coordinates of the vector (1, 1, 1) with respect to the basis {(1, 1, 1), (0, 1, 1,), (0, 0, 1)}? What are its coordinates with respect to the standard basis {(1, 0, 0, (0, 1, 0,), (0,...
-
Evaluate how many lines there are in a true rotational spectrum of CO molecules whose natural vibration frequency is w = 4.09 1014 s1 and moment of inertia I = 1.44 1039 g cm2.
-
The wavenumber of the incident radiation in a Raman spectrometer is 20 623 cm 1 . What is the wavenumber of the scattered Stokes radiation for the J=42 transition of 16 O 2 ?
-
Use appropriate electronic structure software to perform calculations on H 2 O and CO 2 with basis sets of your or your instructors choosing. (a) Compute ground-state energies, equilibrium geometries...
-
A 5.00mm cell was filled with a solution of a dye. The concentration of the dye was 18.5mmol dm 3 . Calculate the molar absorption coefficient of the dye at this wavelength given that the...
-
Alec was an actor and producer in a small, independent film. He had a scene coming up where he had to shoot an actor, Miranda. He organised a professional shooter to come to the film set to give him...
-
What is marketing and why is it important? (details and full explanation)
-
Develop 6 questions (2 per code) that you would ask a BCBA who recommended the system to you, a parent, for their child. 2.14 Selecting, Designing, and Implementing Behavior-Change Interventions...

Study smarter with the SolutionInn App


