The ClO radical decays rapidly by way of the reaction, 2 ClO Cl 2 + O
Question:
The ClO radical decays rapidly by way of the reaction, 2 ClO → Cl2 + O2. The following data have been obtained:
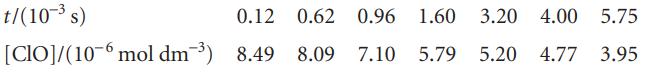
Determine the rate constant of the reaction and the half-life of a ClO radical.
Transcribed Image Text:
t/(10-³ s) [CIO]/(10-6 mol dm³) 8.49 8.09 7.10 5.79 5.20 0.12 0.62 0.96 1.60 3.20 4.00 5.75 4.77 3.95
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Given From the tableplot Concentration Vs time as shown in table for different order reactions the d...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The CIO radical decays rapidly by way of the reaction, 2 CIO ---7Cl, + Oz. The following data have been obtained: Determine the rate constant of the reaction and the half-life of a CIO radical. t/(10...
-
Following data have been obtained for machining AA390 Aluminum, a Si-Al alloy. Compute the K and n values for the Taylor tool life equation. How do these n values compare to the typical values?...
-
The following paired sample data have been obtained from normally distributed populations. Construct a 90% confidence interval estimate for the mean paired difference between the two population...
-
What are the Key Success factors in the Daycare industry? explain
-
Long-time friends Pat and Tom agree on many things, but not the outcome of the American League pennant race and the World Series. Pat is originally from Boston, and Tom is from New York. They have a...
-
The treasurer of Amaro Canned Fruits, Inc., has projected the cash flows of Projects A, B, and C as follows: Suppose the relevant discount rate is 12 percent per year. a. Compute the profitability...
-
A European recession and the U.S. economy a. In 2014, European Union spending on U.S. goods accounted for \(18 \%\) of U.S. exports (see Table 17-2), and U.S. exports amounted to \(15 \%\) of U.S....
-
Accounting for bonds using amortized cost measurement based on the historical market interest rate. Several years ago, Huergo Dooley Corporation (HDC) issued $2,000,000 face value, 8% semiannual...
-
State suitable case for each model Hierarchical Model Network data model Relational model Object-oriented data model
-
Poindexters electric stove short circuited out and a fire ensued. The flames spread over the stove top and onto adjacent countertops eventually destroying wooden storage cabinets on either side of...
-
At 518C, the rate of decomposition of a sample of gaseous acetaldehyde, initially at a pressure of 363 Torr, was 1.07 Torr s 1 when 5.0 per cent had reacted and 0.76 Torr s 1 when 20.0 per cent had...
-
The rate law for the reaction in Exercise 22.2a was reported as d[C]/dt = k[A][B][C]. Express the rate law in terms of the reaction rate; what are the units for k in each case? Data in Exercise 22.2a...
-
Why is knowledge of auditing valuable to an accountant who is not working as an auditor?
-
In 2012, some economists and policymakers continued to worry about the state of the European financial system. An article in the Economist magazine commented on the actions of the European Central...
-
An article in the Wall Street Journal noted that new federal banking regulations mean only a 5 percent reduction in [bank] revenue from households with $500,000 in assets or more compared with a 35...
-
In 2012, an article in the Wall Street Journal described how Amazon had begun making loans to the small businesses that sell on its site. Many of these businesses had previously been turned down for...
-
Suppose that Bank of America pays a 2% annual interest rate on checking account balances while having to meet a reserve requirement of 10%. Assume that the Fed pays Bank of America an interest rate...
-
Evaluate the following statement: The United States has more than 6,000 banks, while Canada has only a few. Therefore, the U.S. banking industry must be more competitive than the Canadian banking...
-
A recent study ("Snack Ads Spur Children to Eat More," The New York Times, July 20, 2009, p. B3) found that children who watched a cartoon with food advertising ate, on average, 28.5 grams of...
-
Write out the formula for the total costs of carrying and ordering inventory, and then use the formula to derive the EOQ model. Andria Mullins, financial manager of Webster Electronics, has been...
-
Consider the following mechanism for renaturation of a double helix from its strands A and B: Derive the rate equation for the formation of the double helix and express the rate constant of the...
-
Methane is a by-product of a number of natural processes (such as digestion of cellulose in ruminant animals, anaerobic decomposition of organic waste matter) and industrial processes (such as food...
-
T. Gierczak, R.K. Talukdar, S.C. Herndon, G.L. Vaghjiani, and A.R. Ravish Ankara (J. Phys. Chem. A 101, 3125 (1997)) measured the rate constants for the bimolecular gas-phase reaction of methane with...
-
Togo's Sandwiches acquired equipment on April 1, 2024, for $18,000. The company estimates a residual value of $2,000 and a five- year service life. Required: 1. Calculate depreciation expense using...
-
1. The ionic solid (NH 4 ) 2 SO 4 is a strong electrolyte and dissolves 100% in aqueous solution. If 52.9 g solid (NH 4 ) 2 SO 4 dissolves into 100 mL water, please answer the following three...
-
Garden Depot is a retailer that is preparing its budget for the upcoming fiscal year. Management has prepared the following summary of its budgeted cash flows: Total cash receipts Total cash...

Study smarter with the SolutionInn App


