The concentration dependence of the osmotic pressure of solutions of a macromolecule at 20C was found to
Question:
The concentration dependence of the osmotic pressure of solutions of a macromolecule at 20°C was found to be as follows:
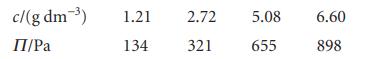
Determine the molar mass of the macromolecule and the osmotic virial coefficient.
Transcribed Image Text:
c/(g dm-3) П/Pa 1.21 134 2.72 321 5.08 655 6.60 898
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Solution P RV NB where N is the number of particles in the solution B is a consta...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A major airline manufacturer was found to be in violation of FAA safety rules and was forced to install additional safety devices in each of its planes within the next six months. The airline company...
-
A major airline manufacturer was found to be in violation of FAA safety rules and was forced to install additional safety devices in each of its planes within the next six months. The airline company...
-
A major airline manufacturer was found to be in violation of FAA safety rules and was forced to install additional safety devices in each of its planes within six months. The airline company projects...
-
Which statement does NOT reflect a way that journals require authors to disclose potential or actual conflicts of interest? Group of answer choices Require researcher's Federal tax statements Require...
-
The following hypotheses are to be tested: H0: p 0.65 HA: p > 0.65 A random sample of 500 is taken. Using each set of information following, compute the power of the test. a. = 0.01, true p = 0.68...
-
According to the Bureau of Labor Statistics, the average workweek for an adult in the United States in April 2018 was 34.5 hours. Assume the population standard deviation for the number of hours...
-
Consider the Gallo strategic decision. Describe how you would go about evaluating that decision.
-
Carole Dahl is chief financial officer of the Phoenix Division of Dahl Corporation, a multinational company with three operating divisions. As part of the budgeting process, Dahl's staff is...
-
What will be the output from the following code? void myFunction(int &b){ } b = 1; int main(){ int a = 0; cout < < a < < " "; myFunction(a); cout < < a; return 0; }
-
After flying with United Airlines, passengers were asked to record their perceptions regarding their experience pertaining to each of the decisionmaking criteria The results are displayed in Table...
-
It is observed that the critical micelle concentration of sodium dodecyl sulfate in aqueous solution decreases as the concentration of added sodium chloride increases. Explain this effect.
-
What is the relative rate of sedimentation for two spherical particles of the same density, but which differ in radius by a factor of 10?
-
Consider the material in Chapter 11 on computer crime and information technology security. a. Which business risks / threats impact Big Marker? Explain your response. b. How does the narrative...
-
What is 'judgement', and how does it apply in business? Give three examples.
-
The manager of a store recorded the annual sales S (in thousands of dollars) of a product over a period of 7 years, as shown in the table, where t is the time in years, with t = 8 corresponding to...
-
Explain the importance of trends to analyse business performance. What methods and indicators would you use to analyse performance trends?
-
Describe how accounting information is used in each of the stages of problem solving.
-
Explain what is meant by the lower-of-cost-or-Net Realisable value method.
-
What are two of the main provisions of FLSA?
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
A molecule in a gas undergoes about 1.0 X 109 collisions in each second. Suppose that (a) Every collision is effective in deactivating the molecule rotationally and (b) That one collision in 10 is...
-
Calculate the frequency of the J = 3 f-- 2 transition in the pure rotational spectrum of 12CI60. The equilibrium bond length is 112.81 pm.
-
If the wave number of the J = 1 f-- 0 rotational transition of IH81Br considered as a rigid rotator is 16.93 cm-1, what is (a) The moment of inertia of the molecule, (b) The bond length?
-
Discussion Topic Initial Reply (5 points) Part 1: In 2-3 sentences and in your own words, explain what do measures of variability measure? Part 2: Create a data set (either on a interval or ratio...
-
Information related to Runner Limited is presented below for its first month of operations. Credit Sales Cash Collections Jan. 7 Chiu Corp. $1,720 Jan. 17 Chiu Corp. $640 15 Elbaz Inc. 6,240 24 Elbaz...
-
1. For the following 2-layer feedforward neural network, compute the outputs for the following sets of inputs: (x=0, x2=0), (x=1, x2=0), (x=0, x2=1), and (x1=1, x2=1). The activation function is also...

Study smarter with the SolutionInn App


