The following data are for the chemisorption of hydrogen on copper powder at 25 C. Confirm that
Question:
The following data are for the chemisorption of hydrogen on copper powder at 25 °C. Confirm that they fit the Langmuir isotherm at low coverages. Then find the value of α for the adsorption equilibrium and the adsorption volume corresponding to complete coverage.
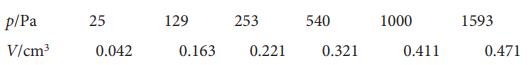
Transcribed Image Text:
p/Pa V/cm³ 25 0.042 129 0.163 253 0.221 540 0.321 1000 0.411 1593 0.471
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Calculations The Langmuir isotherm is given by PP01PP0 where fractional coverage P pressure ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The following data are for Ernst Company. Note: All inventory is purchased on account, and Accounts Payable relates only to the purchase of inventory. Instructions: Compute the following: 1. The...
-
The following data are for a simple steam power plant as shown in Fig. P6.39 State 1 2 3 4 5 6 7 P MPa 6.2 6.1 5.9 5.7 5.5 0.01 0.009 T C 45 175 500 490 40
-
The following data are for a simple steam power plant as shown in Fig. P6.39 State 2 3 4 5 P psia 890 830 1.4 900 860 800 1.5 TF 115 350 920 900 110 6.
-
Telstar Limited was incorporated on 2 January 2015. On 3 January 2015, the company ordered plant from Germany at a cost of E1 million. The plant was loaded free on board in Hamburg on 1 March 2015,...
-
Data set: Tennis Students on the college tennis team were interested in knowing if string tension affected the speed and accuracy of a tennis ball. They had recently read an article in the Journal of...
-
The cash book (bank columns) of Ella for the month of May 207 were as follows: Ellas bank statement for May 207 showed the following: The following errors were discovered: 1. Ella had overcast the...
-
At 3:00 a.m. on November 22, 2010, 16-year-old Sydney McLemore was driving a Mazda3 with her friend, Natalie Hurst, in the front passenger seat. The vehicle was traveling south on Ross Bridge Highway...
-
Southwestern University (SWU), a large state college in Stephenville, Texas, 30 miles southwest of the Dallas/Fort Worth metroplex, enrolls close to 20,000 students. In a typical town-gown...
-
You would like your retirement fund to have 450,000$ after 32 years. The bank offers you an APR of 2.6%, compounded quarterly. How much should your quarterly deposits be in order to reach that goal?...
-
Enthalpy of steam at 75 kPa and 573 K is 3075 kJkg referred to liquid water at 273 K If the mean heat capacity of liquid water and water vapor are 42 and 197 kJkg K respectively, calculate the heat...
-
Drawing from knowledge you have acquired through the text, describe the advantages and limitations of each of the microscopy, diffraction, and ionizations techniques designated by the acronyms AFM,...
-
The volume of oxygen gas at 0 C and 104 kPa adsorbed on the surface of 1.00 g of a sample of silica at 0 C was 0.286 cm 3 at 145.4Torr and 1.443 cm 3 at 760Torr. What is the value of V mon ?
-
Titusberry Inc. is considering the introduction of a new product. Based on past experience, management believes that the distribution of sales demand is normal. Management's best guess is that the...
-
The questions below should be calculated based on the following assumptions (which will be the same for questions 14-17) : The band Thrice consists of four members: Dustin Kensrue, Teppei Teranishi,...
-
The 80/20 principle in fashion means that: Group of answer choices 80% of your sales will be the most important product the smallest portion of product styles will lead to the largest portion of...
-
DomCo, a domestic corporation, is a wholly-owned subsidiary of its Japanese parent corporation, ASIAco, which manufactures lightbulbs. During the current year, DomCo pays $100 million for 100 million...
-
Use these materials: Use the "Sources of Law" section of the Lau & Johnson (pages. 14-22) This is the website for "Sources of Law"...
-
1. What would I find on a job-cost sheet; please give examples? 2. In your own words explain to your classmates what Job Costing is. 3. Explain a job-order costing system and give examples? 4. What...
-
Prove that each of the following functions is uniformly continuous on (0, 1). (You may use l'Hopital's Rule and assume that sin x and log x are continuous on their domains.) a) b) c) f(x) = x log x...
-
In Exercises 1-2, rewrite each verbal statement as an equation. Then decide whether the statement is true or false. Justify your answer. 1. The logarithm of the difference of two numbers is equal to...
-
Describe the model used to determine the heat capacity of atomic crystals.
-
What is the Boltzmann formula, and how can it be used to predict residual entropy?
-
How does the Boltzmann formula provide an understanding of the third law of thermodynamics?
-
2. Suppose that a convolutional layer of a neural network has an input tensor X and computes an output via correlation and ReLU activation, i.e., Z[i, m, j1, j2] =W[m, n, k, k2]X[i, n, j1 + k1, j2...
-
Consider a relation with schema R (A, B, C, D) and FDs the set F: F={ } AB A C C D a. (5%) Determine the set of all candidate keys for the relation schema. b. (5%) Determine the highest normal form...
-
Given the following figure and information, solve for the following: A. The working matrix of the system of differential equations; B. The mathematical model for the amt. of salt in each of the tanks...

Study smarter with the SolutionInn App


