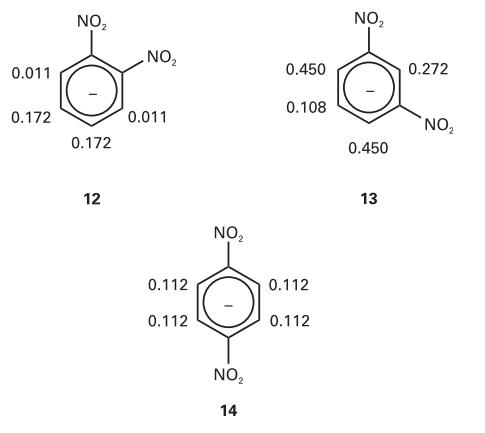
The hyperfine coupling constants observed in the radical anions (12), (13), and (14) are shown (in millitesla,
Question:
The hyperfine coupling constants observed in the radical anions (12), (13), and (14) are shown (in millitesla, mT). Use the value for the benzene radical anion to map the probability of finding the unpaired electron in the π orbital on each C atom.
Transcribed Image Text:
0.011 0.172 NO ₂ 0.172 12 NO₂ 0.011 0.112 0.112 NO ₂ NO ₂ 14 0.450 0.108 0.112 0.112 NO₂ 0.450 13 0.272 NO₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Solution For radical anions hyperfine coupling constants are give...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The hyperfine coupling constant in CH3 is 2.3 mT. Use the information in Table 15.3 to predict the splitting between the hyperfine lines of the spectrum of CD what are the overall widths of the...
-
The hyperfine interaction in a hydrogen atom between the magnetic dipole moment of the proton and the spin magnetic dipole moment of the electron splits the ground level into two levels separated by...
-
The relative orbital levels for the hydrogen atom can be represented as Draw the relative orbital energy levels for atoms with more than one electron, and explain your answer. Also explain how the...
-
Prove that, the area of the traverse is equal to the algebraic sum of the products of the total latitude of each point and algebraic sum of the departures of the lines meeting at that point.
-
Discussion Questions: 1. After returning from the training session at Hamburger University, a McDonalds store owner selected a random sample of 362 drive-thru customers and carefully measured the...
-
The 0.5-kg flyballs of a centrifugal governor revolve at a constant speed v in the horizontal circle of 150-mm radius shown. Neglecting the mass of links AB, BC, AD, and DE and requiring that the...
-
A recent survey of the Youth Health Information Desk at the Catholic University of the Sacred Heart, Rome, enquired about health awareness among students. The respondents voluntarily attended classes...
-
Consider a pressure surge system to reduce the effect of pressure variations at a compressor outlet on the pressure in a compressed gas header. We want to develop a two-tank model and evaluate the...
-
In reactive distillation, how does the integration of chemical reactions within the distillation process affect equilibrium and kinetics? Discuss applications where this technique is particularly...
-
Choke Cherry Ltd. is a private company reporting under ASPE. Its adjusted trial balance at its fiscal year end, December 31, 2014, is shown below: Instructions (a) Prepare an income statement,...
-
Sketch the appearance of the 1 H-NMR spectrum of acetaldehyde (ethanal) using J = 2.90 Hz and the data in Exercise 15.9a in a spectrometer operating at (a) 250 MHz, (b) 500 MHz. Data in Exercise...
-
The chemical shift of the CH 3 protons in acetaldehyde (ethanal) is = 2.20 and that of the CHO proton is 9.80. What is the difference in local magnetic field between the two regions of the molecule...
-
Predict the organic product from each of the following reduction reactions. (a) (b) (c) OH NaBH (1) LiAIH (2) aq. H2SO NaBH
-
ces Primare Corporation provided the following data for last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor Manufacturing overhead...
-
What is the difference between sale of assets and the sale of stocks? How would you Compare the sale of assets and the sale of stocks.
-
Chesterfield Company holds cash of $61,000, inventory worth $112,000, and a building worth $141,000. Unfortunately, the company also has accounts payable of $191,000, a note payable of $91,000...
-
What sort of buyer strategic or financials is more likely to give a bigger purchase premium? Explain.
-
What kind of synergies revenue or cost have the best chance of being realized? Explain
-
Ionization measurements show that a particular lightweight nuclear particle carries a double charge (= 2e) and is moving with a speed of 0.710c. Its measured radius of curvature in a magnetic field...
-
Write a program to move a signed number from smaller register to bigger register. Hint: movzx ax, bl Topic: Data Related Operators and Directives in assembly language
-
The dissociation vapour pressure ofNH4Cl at 427C is 608 kPa but at 459C it has risen to 1115 kPa. Calculate (a) The equilibrium constant, (b) The standard reaction Gibbs energy, (c) The standard...
-
Estimate the temperature at which CuS045H,O undergoes dehydration.
-
For PbI2(s) = 0Pb+(aq) + 2 r(aq), K = 1.4 X 10-8 at 25C and the standard Gibbs energy of formation ofPbI2(s) is -173.64 k] mol ". Calculate the standard Gibbs energy of formation of PbI2 (aq).
-
Superior Company provided the following data for the year ended December 31 (all raw materials are used in production as direct materials): Selling expenses Purchases of raw materials Direct labor...
-
Pietro Frozen Foods, Inc., produces frozen pizzas. For next year, Pietro predicts that 50,000 units will be produced, with the following total costs: Direct materials Direct labor Variable overhead...
-
How does social control operate within online and digital environments, and what challenges arise in regulating behavior in virtual spaces, where traditional mechanisms of control may be less...

Study smarter with the SolutionInn App


