The lowest four energy levels for atomic vanadium (V) have the following energies and degeneracies: What is
Question:
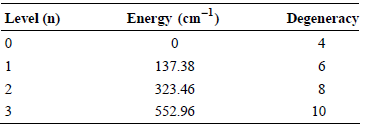
What is the contribution to the average energy from electronic degrees of freedom for V when T = 298 K?
Transcribed Image Text:
Level (n) Energy (cm-1) Degeneracy 4 137.38 6 323.46 552.96 3 10
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Converting to J U N 143 cm 1 hc nN A 143 cm 1 662610 34 j s 30010 10 cm s 1 n ...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Why do electronic degrees of freedom generally not contribute to the constant volume heat capacity?
-
The three lowest energy levels for atomic carbon (C) have the following energies and degeneracies: Determine the electronic contribution to C V for atomic C at 100. K. Level (n) Energy (cm)...
-
The three lowest energy levels for atomic carbon (C) have the following energies and degeneracies: What is the contribution to the average molar energy from the electronic degrees of freedom for C...
-
A restaurant records the following data over a month for its food: Opening inventory: $31,000 Purchases: $88,000 Closing Inventory: $28,000 Transfers in: $800 Transfers out: $200 Employee meals:...
-
In Exercises 1-2, find the center, foci, and vertices of the hyperbola. Then sketch the hyperbola using the asymptotes as an aid 1. (x - 2)2 / 16 - (y + 1)2 / 9 = 1 2. (x - 1)2 / 144 (7 - 4)2 / 25 = 1
-
A rare French cheese sells for 60.00 euros per pound. Assuming a very similar cheese in Wisconsin sells for $55.00 per pound, what is the exchange rate between the euro and the U.S. dollar according...
-
For each of the following, (a) calculate the mean, standard deviation, and standard error of the mean for each Group and (b) construct a bar graph (assume the independent variable is condition and...
-
A car dealer acquires a used car for $ 14,000, terms FOB shipping point. Additional costs in obtaining and offering the car for sale include $ 250 for transportation-in, $ 900 for import duties, $...
-
Royal International Bank (RIB) charges an interest rate of 1.25% per month on loans to its customers. Like all lenders, RIB must report an annual percentage rate (APR) to customers. What should the...
-
"A producer of computer aided design software for the aerospace industry receives numerous calls for technical support. Tracking software is used to monitor response and resolution times. In...
-
Consider an ensemble of units in which the first excited electronic state at energy ε 1 is m 1 -fold degenerate, and the energy of the ground state is m0 -fold degenerate with energy...
-
Consider the following table of diatomic molecules and associated rotational constants: a. Calculate the rotational temperature for each molecule. b. Assuming that these species remain gaseous at 100...
-
Use the Hamiltons Data Set when making the following calculations: a. Compute earnings per share (EPS) for Hamiltons. The number of shares outstanding was 710,000. b. Compute Hamiltons price/...
-
In November 2011, the government of France announced that it was reducing its forecast for economic growth in 2012. It was also reducing its estimates of tax revenue for 2012, since a weaker economy...
-
In the economy of Popania in 2014, total Popanian purchases of assets in the rest of the world equaled $300 billion, purchases of Popanian assets by the rest of the world equaled $400 billion, and...
-
A large electronics firm is attempting to hire six new electrical engineers. It has been the firm's experience that 35 percent of the college graduates who are offered positions with the firm have...
-
No one likes doing chores. In happiness surveys, housework is ranked down there with commuting as activities that people enjoy the least. Maybe thats why figuring out who does which chores usually...
-
Ludlow Inc. started business in 2017. It uses the simplified dollar-value LIFO inventory method. Ending inventories at actual prices are as follows: The price index in 2017, 2018 and 2019 is \(120...
-
Long snorkel. Inhalation of a breath occurs when the muscles surrounding the human lungs move to increase the volume of the lungs, thereby reducing the air pressure there. The difference between the...
-
Accounting policies and practices that are most important to the portrayal of the companys financial condition and results, and require managements most difficult, subjective, or complex judgments...
-
Determine the electron configuration for each of the following atoms: a. Carbon b. Oxygen c. Boron d. Fluorine e. Sodium f. Aluminum
-
In each case, identify the more stable anion. Explain why it is more stable. (a) (b) (c) vs. N. vs. -zo
-
Atropine, extracted from the plant Atropa belladonna, has been used in the treatment of bradycardia (low heart rate) and cardiac arrest. Draw the enantiomer of atropine: CH 0= -
-
Image transcription text Wind tunnel measurements of the pressure and skin friction around a NACA 2415 airfoil at 8 degrees angle of attack resulted in the following data of pressure and skin...
-
Image transcription text The following table contains load-extension data from a tensile test on a cylindrical specimen with gauge length 9mm and gauge diameter 5mm. Load-extension Data Load [KN] 0...
-
Image transcription text Systems Modelling and Analysis - Assignment 1 Due: Friday 25/08/2022 by 5:00:00 pm. To be submitted individually on Canvas and Gradescope. Part 1: Dartboard Positioning...

Study smarter with the SolutionInn App


