The RiceHerzfeld mechanism for the thermal decomposition of acetaldehyde (CH 3 CO(g)) is Using the steady-state approximation,
Question:
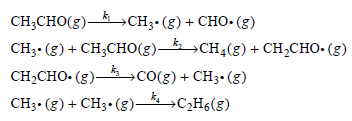
Using the steady-state approximation, determine the rate of methane (CH4(g)) formation.
Transcribed Image Text:
CH CHO(3) — CHз- (3) + CHO- (8) CH- (g) + CH,CHо(з) — сн4(8) + сH-CнO-(3) CH2CHO- (g)– CH- (8) + CHз-(g)—сН6(8) —со() + CHз- (8)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
The differential rate expressions for methane and relevant intermediate species ...View the full answer

Answered By

Shebla K
I am an MBA graduate having experience as an Assistant Professor at University level for two years. I always prepare well for a class as I believe that only if you become an ocean you can give a bucket of water. Being a teacher was not only my profession but also my passion.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For the reaction 2A + B products a friend proposes the following mechanism: A + B M A + M products a. Assuming that the second step is the rate-determining step and the first step is a fast...
-
Because of its importance in atmospheric chemistry, the thermal decomposition of nitric oxide, 2 NO (g) -7 N,(g) + O,(g), has been amongst the most thoroughly studied of gas-phase reactions. The...
-
The thermal decomposition of nitryl chloride, NO2Cl, 2NO2Cl(g) 2NO2(g) + Cl2(g) is thought to occur by the mechanism shown in the following equations: What rate law is predicted by this mechanism?...
-
Harry Bhel carries a business as a sole proprietorship. During its 2022 fiscal period, its first year of operations, the business had cash sales of $123,000. It also has sales on account of $46,000,...
-
Find the zeros of the function algebraically. 1. f(x) = 5x2 + 4x - 1 2. f(x) = 8x + 3/11 - x
-
The boiling point of water on top of a mountain reduced to \(80^{\circ} \mathrm{C}\) compared to \(100^{\circ} \mathrm{C}\) at the sea level. Is the boiling point of water intensive or extensive?
-
Table B. 17 contains hospital patient satisfaction data. Fit an appropriate regression model to the satisfaction response using age and severity as the regressors and account for the medical versus...
-
1. How was mismanagement at Krispy Kreme reflective of leadership failure? 2. Describe the financial shenanigans used by Krispy Kreme. In this regard, is earnings management always a sign of failed...
-
Mike is paid to advise his clients on how to invest their money. One day, he is reviewing the financial statements for a publicly traded company, and believes the company is poised to gain...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
In the troposphere carbon monoxide and nitrogen dioxide undergo the following reaction: NO 2 (g) + CO(g) NO(g) + CO 2 (g) Experimentally, the rate law for the reaction is second order in NO 2 (g),...
-
Reciprocal plots provide a relatively straightforward way to determine if an enzyme demonstrates Michaelis Menten kinetics and to determine the corresponding kinetic parameters. However, the slope...
-
List the properties of the normal density curve.
-
What are some reasons for the current merger wave?
-
Give an example of an impediment to problem definition.
-
What are the four economic types of mergers?
-
How does a nonsymmetric hedge differ from a natural hedge? Provide an example of a nonsymmetric hedge.
-
What is breakup value?
-
In 2020, Pennington Corporation had net sales of $600,000 and cost of goods sold of $360,000. Operating expenses were $153,000, and interest expense was $7,500. The corporations tax rate is 30%. The...
-
A parking lot charges $3 for the first hour (or part of an hour) and $2 for each succeeding hour (or part), up to a daily maximum of $10. (a) Sketch a graph of the cost of parking at this lot as a...
-
A sample of propane (C 3 H 8 ) is placed in a closed vessel together with an amount of O 2 that is 2.15 times the amount needed to completely oxidize the propane to CO 2 and H 2 O at constant...
-
Propose a structure for a compound with molecular formula C 10 H 14 O that exhibits the following 1 H NMR spectrum. Proton NMR Chemical shift (ppm)
-
A van der Waals gas undergoes an isothermal reversible expansion under conditions such that z < 1. Is the work done more or less than if the gas followed the ideal gas law?
-
I need to find out how to calculate the 3-year % rate of return. The information I have is a $10,000 investment and a 12-month yield percentage (1.138 for AB company, 1.894 for AIG company, 0.679 for...
-
Anna has an investment that will bring her $100 with a 30% probability and $40 with a 70% probability. Anna's Utility function is U = Y (1/2) . Where Y= income. Anna is considering selling this...
-
on January 1, 2000, the price of koka kola was $10. on jan and, 2020, the shares were worth $100. the stock Paid no dividends during the period. what is the annual geometric return.

Study smarter with the SolutionInn App


