Use standard Gibbs energies of formation to calculate the standard reaction Gibbs energies at 298K of the
Question:
Use standard Gibbs energies of formation to calculate the standard reaction Gibbs energies at 298K of the reactions
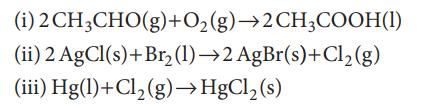
Transcribed Image Text:
(i) 2 CH3CHO(g) + O₂(g) →2CH3COOH(1) (ii) 2 AgCl(s) + Br₂ (1)→2 AgBr(s)+Cl₂(g) (iii) Hg(1)+Cl₂(g) →HgCl₂ (s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Answer and thorough explanation For reaction i we ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Use standard Gibbs energies of formation to calculate the standard reaction Gibbs energies at 298 K of the reactions in Exercise 3.8b.
-
Sandesh and Rani are the only directors and shareholders of SaR Pty Ltd. SaR owns several tenanted properties. Most of the properties are commercial buildings, but it also owns some residential...
-
Use the tabulated free energies of formation to calculate the equilibrium constant for the reaction at 298 K: H 2 (g) + Cl 2 (g) 2HCl (g)
-
Subsequent Events and Subsequently Discovered Facts. Michael Ewing is auditing the financial statements of Dallas Company for the year ended December 31, 2014. In concluding the process of gathering...
-
On January 1, 2015, the Tara Clothing Corporation issued $ 900,000 par value, 5%, six- year bonds. Interest is payable semiannually each January 1 and July 1 with the first interest payment due at...
-
The uniform slender bar of mass p per unit length is freely pivoted about the y-axis at the clevis, which rotates about the fixed vertical z-axis with a constant angular velocity w. Determine the...
-
Oncology Tech is a manufacturer of cancer therapy devices called compensators. CMS was a company that sold cancer treatment planning software for use in purchasing cancer treatment devices. In 2006,...
-
In response to a growing awareness of gluten allergies, Singletary Cookie Company tried using gluten-free flour in its three most popular cookies. After several attempts and a lot of inedible...
-
The KLM Partnership, which uses the accrual method of accounting, is owned equally by Karen ( cash method taxpayer ) , and LM corporation ( accrual method taxpayer ) . Karen is a real estate...
-
National Foods is considering producing a new candy, Nasty-As- Can-Be. National has spent two years and $450,000 developing this product. National has also test marketed Nasty, spending $100,000 to...
-
Discuss the relationships between the various formulations of the Second Law of thermodynamics.
-
Use mathematical software, a spreadsheet, or the Living graphs on the web site for this book to: (a) Evaluate the change in entropy of 1.00mol CO 2 (g) on expansion from 0.001m 3 to 0.010m 3 at 298K,...
-
How can a target zone help create a more stable exchange rate? Explain.
-
Jane Evans earned $2,000A deluxe cheeseburger is purchased at a Chief's game for $22.00, which includes the sales tax. The sales tax rate is 7.00%. What will be the amount sales tax that will need to...
-
How can you highlight compnents of a formula when working inside a cell? Group of answer choices Ctrl Shift $ Ctrl Shift Enter Ctrl Shift ( ( Arrow ) )
-
For the current year, Bell Corporation had worldwide taxable income of $675,000 and a tentative United States income tax of $229,500. Bell's taxable income from business operations in Country A was...
-
Consider the curve described by x = -3t+ 2, y = 5t-4 = -1 and to = 1. with the points P and Q defined by the points on the curve where tp What is the length of the curve between points P and Q in...
-
Write an introduction for essay exploring the 10K Annual report of Amazon and its biggest competitor, Walmart, and comparing the two and answering general essay questions involving internal controls,...
-
NASA astronauts retrieved a satellite and brought it into the cargo bay of the space shuttle, as shown in Figure E10.7(a). A model of the feedback control system is shown in Figure. E10.7(b)....
-
The roof of a refrigerated truck compartment is of composite construction, consisting of a layer of foamed urethane insulation (t2 = 50 mm, ki = 0.026 W/m K sandwiched between aluminum alloy panels...
-
Discuss the steps involved in the calculation of the energy of a system by using the variation principle.
-
Hydrogen is the most abundant element in all stars. However, neither absorption nor emission lines due to neutral hydrogen are found in the spectra of stars with effective temperatures higher than 25...
-
Give the possible term symbols for (a) Li [He]2s 1 , (b) Na [Ne]3p 1 .
-
You'll be arguing against this new energy bar. How do you find your supporting evidence for your argument? A. Visit online medical journals with studies related to energy supplements and the...
-
Wayfair produces four types of frames. Each type requires a certain amount of skilled labor, metal, and glass, as shown in the table below. The unit selling price of each type is also shown in the...
-
You are a chief of a large city agency. Your department is working with the Federal government through the Secure Communities program. Your city has been declared a sanctuary city and has developed a...

Study smarter with the SolutionInn App


