According to Figure 1, a Fauna H rock would most likely form under which of the following
Question:
According to Figure 1, a Fauna H rock would most likely form under which of the following pressure and temperature conditions?
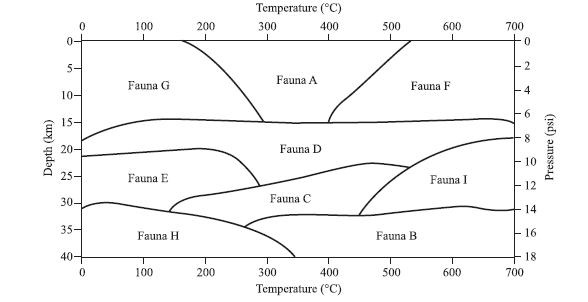
Pressure Temperature
A. 4 psi 200◦C
B. 8 psi 500◦C
C. 12 psi 350◦C
D. 16 psi 150◦C
Transcribed Image Text:
Depth (km) 0 10- 15- 20- 25- 30- 35- 40- 0 100 1 Fauna G Fauna E Fauna H T 100 200 200 Temperature (°C) 400 300 Fauna A 300 Fauna D Fauna C 400 Temperature (°C) 500 Fauna B 500 600 Fauna F Fauna I 600 700 1 1 0 I 2 SO 10 12 +14 - 16 18 m 700 Pressure (psi)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
The best answer is D To answer this question you must ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Business questions
-
Cast fossils are formed when material is placed into a depression in a rock near the surface. They are formed at very high temperatures and low pressures. According to Figure 1, cast fossils are most...
-
Under which of the following reaction conditions would (R)-2-chlorobutane form the most (R)-2-butanol: HO- in 50% water and 50% ethanol or HO- in 100% ethanol?
-
1. Hannah is applying for a life policy on her girlfriend Sarahs life. The policy is $500,000 and carries a large premium. Hannah is the main earner, so she is concerned about not being able to pay...
-
Briefly explain the differences between copyrights and patents.
-
SolarTech Company manufactures a special lacquer, which is used in the aeronautical and space industries. Two departments are involved in the production process. In the Mixing Department, various...
-
For the MIPS assembly instructions below, what is the corresponding C statement? Assume that the variables f, g, h, i, and j are assigned to registers $s0, $s1, $s2, $s3, and $s4, respectively....
-
Use the financial data for Randa Merchandising, Inc., in Exercise 13-13 to prepare its income statement for calendar-year 2017. (Ignore the earnings per share section.) Data From Exercise 13.13 In...
-
In Shady Company, materials are entered at the beginning of each process. Work in process inventories, with the percentage of work done on conversion costs, and production data for its Sterilizing...
-
1. A hollow conducting sphere has an inner and outer radius of R and R2 respectively. A point charge of +Q sits inside the hollow sphere at the center. Additionally, a charge of +2Q is distributed...
-
Which of the following graphs best represents the relationship between time elapsed and saturation of the rocks observed in Experiment 1? Experiment 1 In a laboratory, a scientist wanted to determine...
-
Afifth colony of Tenebrio molitor was raised on barley, another whole grain, in the same lab. Assuming this colony behaved similarly to the other colonies raised on whole grains, what was most likely...
-
Determine the enthalpy change h of oxygen, in Btu/lbm, as it is heated from 800 to 1500 R, using (a) The empirical specific heat equation as a function of temperature (Table A2Ec), (b) The cp value...
-
Entity A reported the following basic earnings per share and diluted earnings per share: Basic EPS Diluted EPS. 20x5 $0.90 0.85 20x4 $1.00 0.80 Explain the possible reason(s) for basic earnings per...
-
The W.C. Pruett Corp. has $600,000 of interest-bearing debt outstanding, and it pays an annual interest rate of 7%. In addition, it has $600,000 of common equity on its balance sheet. It finances...
-
Marcus owned 200 shares (100 percent) of Sterling Company's common stock. His basis in the shares was \(\$ 12,000\). In 2015 , Sterling issued a preferred stock dividend to Marcus. The 50 shares he...
-
You are completing the audit of Toledo Ltd. for the financial year ended December 31, 2023. Under the terms of a major loan contract, Toledo is required to maintain certain financial ratios. If the...
-
Mr. Garcia, a cashbasis taxpayer, owns an apartment building. His records reflect the following information for 2018: What is the amount of gross rental income Mr. Garcia should include in his gross...
-
When the parent has used the equity method, its net income equals consoli dated net income attributable to its shareholders, and its retained earnings equal consolidated retained earnings. However,...
-
Wilsons Auto Repair ended 2011 with Accounts Receivable of $85,000 and a credit balance in Allowance for Uncollectible Accounts balance of $11,000. During 2012, Wilsons Auto Repair had the following...
-
Cite a local example of an oligopoly, explaining why it is an oligopoly.
-
Distinguish between the following pairs of items that appear on operating statements: (a) Gross sales and net sales, (b) Purchases at billed cost and purchases at net cost.
-
How does gross margin differ from gross profit? From net profit?
-
2022 MFJ, husband age 47 W2: $90K/$9185 tax withheld, wife 45, housewife, not a business person, paints as a hobby and sold a painting for $3K and had the following expenses: supplies: $800 and small...
-
XYZ Company bought rugs from the manufacturer at $50 each to be sold to the customers. The store's overhead expenses are 30% of cost, and the owners require a profit of 20% of cost. (a) For how much...
-
How can the auditor be sure that the sample they have chosen is representative of the population? What would be the impact on the auditor's judgement of a sample that is not representative of the...

Study smarter with the SolutionInn App


