Cathodic stripping of ClO -4 in Figure 17-26 does not involve oxidation or reduction of ClO -4
Question:
Cathodic stripping of ClO-4 in Figure 17-26 does not involve oxidation or reduction of ClO-4. Explain how this measurement works.
In Figure 17-26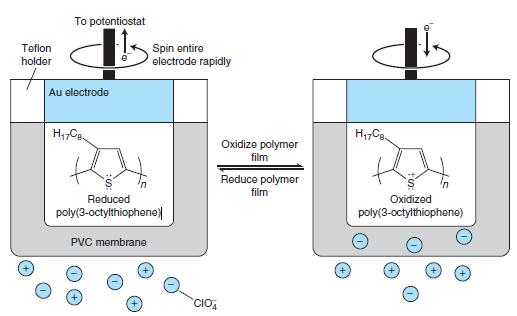
Transcribed Image Text:
To potentiostat Teflon Spin entire electrodo rapidly holder Au electrode Oxidize polymor film Reduce polymer film Reduced Oxidized poly(3-octylthiophene) poly(3-octylthiophene) PVC membrane CIO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
An electrode is dipped into the solution The electrode is connected to a power supply and a current ...View the full answer

Answered By

Jemima Wangu
I am a proficient tutor with the following skills;
Effective time management
Microsoft Office
Computer skills
Multitasking skills
Strong Mathematical skills
Honesty and integrity
Advanced technology skills
Motivated attitude
Content writing
Content research and development
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain why a census does not necessarily have to involve a population of people. Use an example to illustrate.
-
Explain how calorimetry works to calculate H or E for a reaction. Does the temperature of the calorimeter increase or Decrease for an endothermic reaction? How about for an exothermic reaction?...
-
Does this reduction appear to be constant, accelerating, or decelerating? Explain.
-
Divalent carbon species called carbenes care capable of fleeting existence. For example, methylene: CH2, is the simplest carbene. The two unshared electrons in methylene can be either spin-paired in...
-
Suppose you were asked to enter a debate in which your task was to argue against any special effort to manage workforce diversity. What would you say?
-
Write the first five terms of each sequence in Problems 2126. an || (-1)"+1 2"
-
You drop a ball from rest from a window \(12 \mathrm{~m}\) above the ground, and just before it hits the ground its speed is recorded to be \(14.6 \mathrm{~m} / \mathrm{s}\). What percentage of the...
-
Artero Corporation, discussed in Problem 9, is a retailer of toy products. The firms management team recently extended the monthly sales forecasts that were prepared for the last three months of 2011...
-
(Similar to Problem 1. but the area is to be found.) If $49.500 has been allocated for walls. a) What are the dimensions of the largest warehouse that can be built? b) What is the floor area of this...
-
A gaseous fuel with a volumetric analysis of 60 percent CH4, 30 percent H2, and 10 percent N2 is burned to completion with 130 percent theoretical air. Determine (a) The airfuel ratio and (b) The...
-
In 1 M NH 3/1 M NH 4 Cl solution, Cu 2+ is reduced to Cu+ near 20.3 V (versus S.C.E.), and Cu + is reduced to Cu(in Hg) near 20.6 V. (a) Sketch a qualitative sampled current polarogram for a solution...
-
Vapor at a pressure of 30.3 mbar from the solid compound pyrazine had a transmittance of 24.4% at a wavelength of 266 nm in a 3.00-cm cell at 298 K. (a) Convert transmittance to absorbance. (b)...
-
Newly purchased automobile tires of a certain type are supposed to be filled to a pressure of 30 psi. Let denote the true average pressure. Find the P-value associated with each of the following...
-
What is budgeting? Explain.
-
The business is trying to develop a system for good financial governance. What are three examples of conventions this business should follow to maintain compliant financial management throughout all...
-
Under which circumstances might a client choose to set QuickBooks Online to never suggest categorizations from a vendor? Explain.
-
Steven has a $20,000 investment in a five-year XYZ Market-Linked GIC. Return on the GIC is based on the performance of the S&P/TSX Composite Index, with a 65% participation rate. The return on the...
-
Assess the preliminary materiality based on the policy assuming moderate risk and satisfactory results of the analytical procedures (your answer should provide only one materiality measure, i.e.,...
-
A cosmic ray particle travels directly over a football field, from one goal line to the other, at a speed of 0.50c. (a) If the length of the field between goal lines in the Earth frame is 91.5 m (100...
-
The test statistic in the NeymanPearson Lemma and the likelihood ratio test statistic K are intimately related. Consider testing H 0 : = 0 versus H a : = a , and let * denote the test statistic...
-
(a) Calculate the pH of a solution prepared by mixing 0.0100 mol of the base B (Kb = 10 = - 2.00) with 0.020 0 mol of BH+Br- and diluting to 1.00 L. First calculate the pH by assuming [B] = 0.0100...
-
Effect of ionic strength on pKa. Ka for the H2PO4- /HPO24- buffer is If you mix a 1:1 mole ratio of H2PO-4 and HPO42- at 0 ionic strength, the pH is 7.20. Using activity coefficients from Table 7-1,...
-
Systematic treatment of equilibrium. The acidity of Al3+ is determined by the following reactions. Write the equations needed to find the pH of Al(ClO4)3 at a formal concentration F. 2 + 2H20-Al(...
-
Kanesha is an entrepreneur and recently opened her first coffee shop, The Coffee Cat. Kanesha pays $4,800 for rent each month, $3,100 for monthly employee payroll, and $1,800 for supplies each...
-
A, a merchant, supplies B, a minor, with the rice necessary for his consumption. B, refuses to pay the price. Can A recover the Price? Why or why not ? Explain
-
The given equation is a polynomial function of the second degree F(x)= 4-3x 3 + 2x 2 + 5x. solve

Study smarter with the SolutionInn App


