Calculate the pH of a 0.010 M solution of each amino acid in the form drawn here.
Question:
Calculate the pH of a 0.010 M solution of each amino acid in the form drawn here.
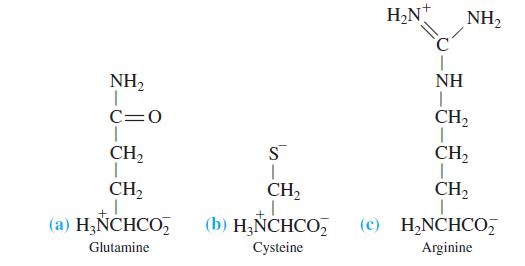
Transcribed Image Text:
H2N NH2 NH2 NH C=0 CH2 CH2 S CH, CH, CH2 CH2 (a) H;NCHCO, (b) H&NCHCO, (c) H,NCHCO, Glutamine Cysteine Arginine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
From the given pH of the each solution 0010M The three f...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Calculate the pH of a 0.42 M NH4Cl solution.
-
Calculate the pH of a 0.15 M aqueous solution of zinc chloride, ZnCl2. The acid ionization of hydrated zinc ion is and Ka is 2.5 10-10.
-
Calculate the pH of a 0.15 M aqueous solution of aluminum chloride, AlCl3. The acid ionization of hydrated aluminum ion is and Ka is 1.4 10-5.
-
Greety Food in Ashland, Kentucky, manufactures and markets snack foods. Sita Lee manages the company's fleet of 220 delivery trucks. Lee has been charged with *reengineering* the fleet-management...
-
Locate IRC Section 162 and answer the following: a. What subsections does it include? b. What paragraphs does Subsection (d) include? c. What subparagraphs does Section 162(d)(3) include?
-
When visibility is good and there are no meteor showers, it is possible to see about 5 to 10 meteors per a. Minute b. Hour c. Night d. Week
-
A US Treasury bond has a maturity of 10 years and a face value of $\$ 10,000$ with a $5 \%$ coupon rate. Trading of the bonds has been pretty active, and the current quoted price is $\$ 96,500$. What...
-
A study of cigarette demand resulted in the following logarithmic regression equation: Here, Q denotes annual cigarette consumption, P is the average price of cigarettes, Y is per capita income, A is...
-
Horton Manufacturing Incorporated produces blinds and other window treatments for residential homes and offices. The owner is concerned about the maintenance costs for the production machinery...
-
At the time of her death, Ariana held the following assets. Fair Market Value Personal residence (title listed as "Ariana and Peter, tenants by the entirety with right of survivorship") $900,000...
-
How many milliliters of 0.800 M KOH should be added to 5.02 g of 1,5-pentanedioic acid (C 5 H 8 O 4 , FM 132.11) to give a pH of 4.40 when diluted to 250 mL?
-
(a) Draw the structure of the predominant form (principal species) of 1,3-dihydroxybenzene at pH 9.00 and at pH 11.00. (b) What is the second most prominent species at each pH? (c) Calculate the...
-
Explain what is meant by the partial fraction decomposition of a rational expression.
-
Consider a discrete-time version of the dynamic Arrow-Debreu-McKenzie model for the same data given as in Exercise E5. Initial prices are: Exercise E5 An owner of a strawberry plantation hires one...
-
An owner of a strawberry plantation hires one worker who has 24 units of time. The employee can allocate part of the time to work and part to rest. He/she owns 20% of shares in profits of the...
-
Why a Hicksian demand function is also called a compensated demand function?
-
What conditions need to be satisfied to have a Hicksian demand function and a Marshallian demand function having the same values?
-
What does it mean that a utility function is a numerical characteristics of a relation of consumers preference?
-
Assume that Marcy Jones is a marketing director in Metro Mobility's head office in Toronto. During 2015 she worked for the company all year at a $6,500 monthly salary. She also earned a year-end...
-
You are interested in investing and are considering a portfolio comprised of the following two stocks. Their estimated returns under varying market conditions are provided: (note: it is difficult to...
-
Using SI units, calculate the change in potential energy of a 150-lb person riding the 15-ft-long uphill portion of a water slide (as described in Problem P3.10). The change in potential energy is...
-
Using the speed given in Problem P3.10, calculate the power required to move the rider in Problem P3.30 up the incline portion of the water slide, where power is the change in energy divided by the...
-
Estimate the force acting on a passengers window in a commercial jet aircraft due to air pressure differential.
-
Describe, in steps, at least one strategy used to attack embedded operating systems. Describe, in steps, at least one strategy used to defend against the chosen attack.
-
how does a hypercompetetice environment challenge those in an industry
-
Describe how managers can reduce unnecessary costs, including real - world examples of how it has been done

Study smarter with the SolutionInn App


