Fractional composition in a diprotic system. Create a spreadsheet with Equations 9-19 through 9-21 to compute the
Question:
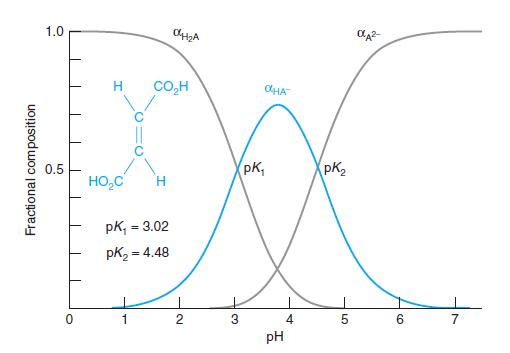
Fractional composition in a diprotic system. Create a spreadsheet with Equations 9-19 through 9-21 to compute the three curves in Figure 9-4. Plot the three curves in a beautifully labeled figure.
Figure 9-4
Equations 9-19 to 9-21

Transcribed Image Text:
1.0 H CO,H OHA pK, PK, 0.5 HO,C pk, = 3.02 %3! pK2 = 4.48 2 3 4 5. 6. 7 pH Fractional composition
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
pH alphaH2A alphaHA alphaA2 0 0999046 0000954 316E08 0070707 0998877 0001123 437E08 0141414 0998679 0001321 606E08 0212121 0998446 0001554 839E08 0282828 0998172 0001828 116E07 0353535 0997849 0002151 ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Fractional composition in a triprotic system. For a triprotic system, the fractional composition equations are where D = [H + ] 3 + K 1 [H + ] 2 + K1K 2 [H + ] + K 1 K 2 K 3 . Use these equations to...
-
Fractional composition in a tetraprotic system. Prepare a fractional composition diagram analogous to Figure 9-4 for the tetraprotic system derived from hydrolysis of Cr3+: Cr3+ + (H2O) Cr(OH)2+ +...
-
A solution containing 0.139 mmol of the triprotic acid tris(2-aminoethyl)amine 3HCl plus 0.115 mmol HCl in 40 mL of 0.10 M KCl was titrated with 0.490 5 M NaOH to measure acid dissociation...
-
Alpha Mann Ltd makes and sells computer carry bags. Bill Blake, the company accountant, is responsible for preparing the company's annual budget. In compiling the budget data for next year, Blake has...
-
Dr. Tooth is a 40-year-old dentist in Small Town, USA. He graduated from dental school five years ago and has had a thriving practice ever since. At the end of last year, however, Dr. Tooth had an...
-
List the three ways in which radio waves from space originate.
-
A material processing oven has a first cost of $\$ 16,999$ with a life of 10 years and a salvage value of $\$ 2,500$. The corporate MARR is $12 \%$. If the system provides about $\$ 2,750$ annually,...
-
When companies venture abroad, managers seek information on the legal and political environments in each country. This information is available from various Web sources, as illustrated in the...
-
(a) Explain the loop problem. Apply Spanning Tree Protocol (STP) in the following network diagram to solve the loop Problem. Find out the Root Bridge, root ports, designated bridge, designated ports,...
-
During a recent management meeting, Steve Micke, director of marketing, proposed that the company begin capitalizing its marketing expenditures as goodwill . In his words, Marketing expenditures...
-
Calculate H2A , HA -, A2- and for cis-butenedioic acid at pH 1.00, 1.92, 6.00, 6.27, and 10.00.
-
Explain what is wrong with the following statement: At its isoelectric point, the charge on all molecules of a particular protein is 0.
-
Environmental Landscaping Inc. is preparing its budget for the first quarter of 2010. The next step in the budgeting process is to prepare a cash receipts schedule and a cash payments schedule. To...
-
Why, in the static Arrow-Hurwicz model, is every allocation which is Pareto optimal and accepted by traders feasible with regard to an initial allocation? Use the Edgeworth box to explain this.
-
What are the mechanisms of reaching an equilibrium state in the Cournot, Stackelberg and Bertrand duopoly models?
-
What are the relationships between a product price set by a monopolist and price elasticity of demand for this product?
-
In each of the following situations, you are given a fact situation plus a rule in the form of a statute. Phrase the legal issue involved in each situation. a. Facts: Len is divorced from Mary, who...
-
Using the Edgeworth box for the static Arrow-Hurwicz model of a market with two traders and two goods present a geometric illustration of a case when there exists no Walrasian equilibrium price...
-
Austin Motors is located in Victoria, British Columbia, and is the only Austin dealer in Western Canada. The dealership repairs and restores Austin vintage cars. Hal Irwin, the general manager, is...
-
Interest Compounded Annually. When P dollars is invested at interest rate i, compounded annually, for t years, the investment grows to A dollars, where A = P(1 + i) t . Trevor's parents deposit $7800...
-
Cable AB on the boom truck is hoisting the 2500-lb section of precast concrete (Figure P4.18). A second cable is under tension P, and workers use it to pull and adjust the position of the concrete...
-
Solve the problem of Example 4.5 by using the force components method. Replace the polar representation of the anchor straps tension by the horizontal and vertical components T x and T y , and solve...
-
A front loader of mass 4.5 Mg is shown in side view as it lifts a 0.75-Mg load of gravel (Figure P4.20). (a) Draw a free body diagram of the front loader. (b) Determine the contact forces between the...
-
What are the main ethical issues that researchers may encounter when conducting business research and how can they address these issues ?
-
How has womens participation in the Olympics changed over the years? Explain
-
What is the Christian view of work according to the God is Not One: The Eight Rival Religions That Run the World.

Study smarter with the SolutionInn App


