What is the purpose of the WO3 WO 3 and Cu Cu in Figure 27-8? Figure 27-8
Question:
What is the purpose of the WO3WO3 and CuCu in Figure 27-8?
Figure 27-8
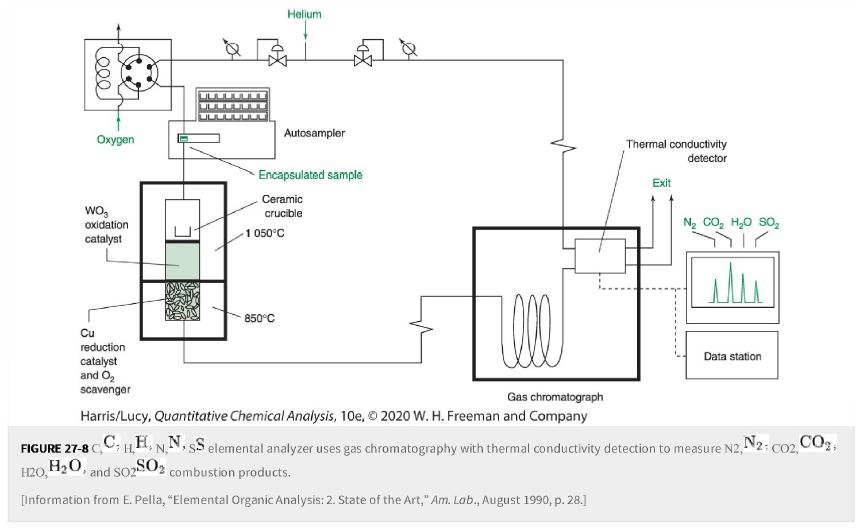
Transcribed Image Text:
Leee Oxygen WO oxidation catalyst Cu reduction catalyst and O₂ scavenger 7. Encapsulated sample Ceramic crucible Helium Autosampler 1 050°C 850°C Well Gas chromatograph Harris/Lucy, Quantitative Chemical Analysis, 10e, © 2020 W. H. Freeman and Company Thermal conductivity detector FIGURE 27-8 C H20, H₂O, and S02 SO2 combustion products. [Information from E. Pella, "Elemental Organic Analysis: 2. State of the Art," Am. Lab., August 1990, p. 28.] Exit Ng CO, HO SO, Sair Data station CHEN. HH, NN, SS elemental analyzer uses gas chromatography with thermal conductivity detection to measure N2, N2: CO2, CO₂,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
The WO3WO3 and CuCu combination is placed in a single process in ord...View the full answer

Answered By

Tamondong Riza
Professionally, I am a teacher with years of experience tutoring math and science, as well as teaching in both public schools and independent schools. I feel that education should be an enlightening experience for all children, and I'm committed to helping my students learn new skills and make progress in their subjects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
What is the purpose of IFRS 8 on Operating Segments?
-
What is the purpose of application controls? How does it differ from the purpose of general controls?
-
What are transportation user charges? What is the purpose of such charges?
-
Elmira, Inc had $20,000,000 of bonds outstanding on Dec 31, 2018. The ten year bonds were issued on Jan 1, 2012 for $21,000,000. Elmira can call the bonds at 102% any time after Jan 1, 2018. At the...
-
Consider a multiple linear regression problem with design matrix Z and observations Y. Let Z1 be the matrix remaining when at least one column is removed from Z. Then Z1 is the design matrix for a...
-
Winter Corporation has just completed its comparative statements for the year ended December 31, 2012. At this point, certain analytical and interpretive procedures are to be undertaken. The...
-
An axial fan operating at \(1000 \mathrm{rpm}\) has the characteristics shown in Fig. P12.42. It delivers \(15{ }^{\circ} \mathrm{C}\) atmospheric air through a 50-cm I.D., galvanized, sheet-metal,...
-
In addition to a Gantt chart, youve drawn Brian a PERT diagram so that you can communicate the necessity to keep an eye on the critical path. Consult Figure which was derived from the data from...
-
The Royal Gorge Bridge in Colorado rises 321 m above the Arkansas River. Suppose you kick a rock horizontally off the bridge. The magnitude of the rock's horizontal displacement is 45.0 m. Find the...
-
Cassi Taxpayer (SSN 412-34-5670) who is single, has a home cleaning business she runs as a sole proprietorship. The following are the results from business operations for the tax year 2014: Gross...
-
Write a balanced equation for combustion of C 8 H 7 NO 2 SBrCl C 8 H 7 NO 2 SBrCl in a C,C,H,H,N,N,SS elemental analyzer.
-
Corrected peak area. A micellar electrokinetic chromatography method for phthalazine and its metabolite 1-1- phthalazinone yielded the following repeatability for six replicate injections of a 50 M...
-
ThyssenKrupp issued the following footnote in the interim financial statements for the first quarter ending December 31, 2007: On the basis of the authorization granted by the Annual Stockholders'...
-
Explain any two (2) methods used to review and modify work practices in a workplace for the workers with different languages
-
SWOT Analysis of domestic violence and Women's Shelter of New York City.
-
Pick any individual multinational business that operates in an overseas market and theoretically review how it has gone about securing its position within that specific market. Attention to should be...
-
Mary did not make any gifts during 2020. John and Mary agreed to split gifts for 2020. What is the amount of gifts that can be split between John and Mary in 2020?
-
Creating structure in any organization is essential for success. An implementation structure for innovation is no different. In this two-part assignment, you will organizational structure for...
-
A researcher is interested in the effect of military service on human capital. He collects data from a random sample of 4000 workers aged 40 and runs the OLS regression Yi = 0 + 1Xi + ui, where Yi is...
-
Prove the following D,(cos x) = - sin x (Hint: Apply the identity cos(A + B) = cos A cos B sin A sin B)
-
(a) Use the ideal gas law (Problem 1-16) to calculate the density (g/mL) of helium at 20C and 1.00 bar. (b) Find the true mass of Na (density = 0.97 g/mL) weighed in a glove box with a He atmosphere,...
-
(a) The equilibrium vapor pressure of water at 20C is 2330 Pa. What is the vapor pressure of water in the air at 20C if the relative humidity is 42%? (Relative humidity is the percentage of the...
-
Effect of altitude on electronic balance. If an object weighs ma grams at distance ra from the center of the Earth, it will weigh mb = ma(r2a/r2b) when raised to rb. An object weighs 100.0000 g on...
-
Reflective writing on the topic Global Trade and Covid-19 is required. Subject: International Economic Issues.
-
How can one efficaciously strategize temporal allocation amidst the multifaceted demands of modern life ? Explain
-
Cruise Mach 0.7 at 35,000 ft, International Standard Atmosphere Fan Pressure ratio: 1.45; Isentropic efficiency 94% LPC Pressure ratio: 1.45; Isentropic efficiency 92%* HPC Pressure ratio: 27.9;...

Study smarter with the SolutionInn App


