Question: What is wrong with this procedure? According to Table 7-1, carbonate can be measured by a Volhard titration. Removal of the precipitate is required. To
What is wrong with this procedure? According to Table 7-1, carbonate can be measured by a Volhard titration. Removal of the precipitate is required. To analyze an unknown solution of Na2CO3,Na2CO3, I acidified the solution with freshly boiled and cooled HNO3 HNO3 to give ~0.5 M HNO3. ~0.5 M HNO3 Then I added excess standard AgNO3, AgNO3 but no Ag2CO3 Ag2CO3 precipitate formed. What happened?
Table 7-1
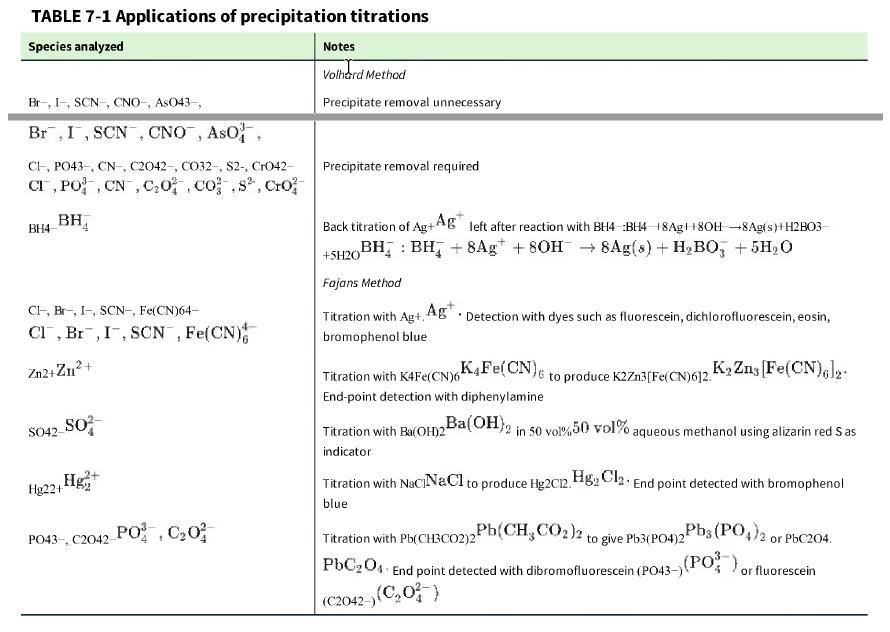
TABLE 7-1 Applications of precipitation titrations Species analyzed Br-, I-, SCN-, CNO-, AsO43-, Br, I, SCN, CNO, ASO, CI, PO43-, CN, C2042-, CO32-, S2-, CrO42- CI, PO, CN, C0, cos, Cro BH4 BH Cl, Br, I-, SCN-, Fe(CN)64- Cl, Br, I, SCN, Fe(CN) Zn2+Zn+ SO42- SO Hg+ Hg22+1 PO43-, C2042-PO, C0 Notes Volhard Method Precipitate removal unnecessary Precipitate removal required Back titration of Ag+Ag left after reaction with BH4 BH4 18Ag8OH-8Ag(s)+H2B03- BH BH + 8Ag +80H 8Ag(s) + HBO3 + 5HO +5H201 Fajans Method Titration with Ag+, Ag. Detection with dyes such as fluorescein, dichlorofluorescein, eosin, bromophenol blue to produce K2Zn3[Fe(CN)612. K Zns [Fe(CN)6]2. Ba(OH)2 in 50 vol%50 vol% aqueous methanol using alizarin red Sas K4Fe(CN)6 Titration with K4Fe(CN)6 End-point detection with diphenylamine Titration with Ba(OH)2 indicator Titration with NaciNaCl to produce Hg2012. HgCl End point detected with bromophenol blue Pb(CH3CO2)2 to give Pb3(PO4)2 Titration with Pb(CH3CO2)2 PbC204 End point detected with dibromofluorescein (PO43-) (C2042-) (C0) Pb3(PO4)2 or PbC204. (PO), or fluorescein
Step by Step Solution
3.40 Rating (163 Votes )
There are 3 Steps involved in it
Thorough explanation The procedure is incorrect because the carbona... View full answer

Get step-by-step solutions from verified subject matter experts


